TheT-cell receptor(TCR) is aprotein complexfound on the surface ofT cells,or T lymphocytes,[1]that is responsible for recognizing fragments ofantigenas peptides bound tomajor histocompatibility complex(MHC) molecules. The binding between TCR and antigen peptides is of relatively lowaffinityand isdegenerate:that is, many TCRs recognize the same antigen peptide and many antigen peptides are recognized by the same TCR.[2]
| TCR complex | |
|---|---|
 | |
| Identifiers | |
| Symbol | TCR |
| OPM superfamily | 166 |
| Membranome | 26 |
| T-cell receptor alpha locus | |
|---|---|
| Identifiers | |
| Symbol | TRA |
| Alt. symbols | TCRA, TRA@ |
| NCBI gene | 6955 |
| HGNC | 12027 |
| OMIM | 186880 |
| Other data | |
| Locus | Chr. 14q11.2 |
| T-cell receptor beta locus | |
|---|---|
| Identifiers | |
| Symbol | TRB |
| Alt. symbols | TCRB, TRB@ |
| NCBI gene | 6957 |
| HGNC | 12155 |
| OMIM | 186930 |
| Other data | |
| Locus | Chr. 7q34 |
| T-cell receptor delta locus | |
|---|---|
| Identifiers | |
| Symbol | TRD |
| Alt. symbols | TCRD, TRD@, TCRDV1 |
| NCBI gene | 6964 |
| HGNC | 12252 |
| Other data | |
| Locus | Chr. 14q11.2 |
| T-cell receptor gamma locus | |
|---|---|
| Identifiers | |
| Symbol | TRG |
| Alt. symbols | TCRG, TRG@ |
| NCBI gene | 6965 |
| HGNC | 12271 |
| Other data | |
| Locus | Chr. 7p14 |
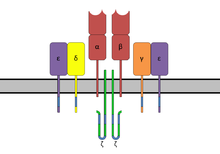
The TCR is composed of two different protein chains (that is, it is aheterodimer). In humans, in 95% of T cells the TCR consists of an alpha (α) chain and a beta (β) chain (encoded byTRAandTRB,respectively), whereas in 5% of T cells the TCR consists ofgamma and delta(γ/δ) chains (encoded byTRGandTRD,respectively). This ratio changes duringontogenyand in diseased states (such asleukemia). It also differs between species.Orthologuesof the 4locihave been mapped in various species.[3][4]Each locus can produce a variety ofpolypeptideswith constant and variable regions.[3]
When the TCR engages with antigenic peptide and MHC (peptide/MHC), the T lymphocyte is activated throughsignal transduction,that is, a series of biochemical events mediated by associated enzymes, co-receptors, specialized adaptor molecules, and activated or releasedtranscription factors.Based on the initial receptor triggering mechanism, the TCR belongs to the family ofnon-catalytic tyrosine-phosphorylated receptors(NTRs).[5]
History
editIn 1982, Nobel laureateJames P. Allisonfirst discovered a clonally expressed T-cell surface epitope in murine T lymphoma.[6]In 1983, Ellis Reinherz first defined the structure of the human T-cell receptor using anti-idiotypic monoclonal antibodies to T-cell clones, complemented by studies in the mouse byPhilippa MarrackandJohn Kappler.[7][8]Then,Tak Wah Mak[9]andMark M. Davis[10]identified the cDNA clones encoding the human and mouse TCR respectively in 1984. These findings allowed the entity and structure of the elusive TCR, known before as the "Holy Grail of Immunology", to be revealed. This allowed scientists from around the world to carry out studies on the TCR, leading to important studies in the fields ofCAR-T,cancer immunotherapyandcheckpoint inhibition.
Structural characteristics
editThe TCR is a disulfide-linked membrane-anchored heterodimeric protein normally consisting of the highly variable alpha (α) and beta (β) chains expressed as part of a complex with the invariantCD3chain molecules. T cells expressing this receptor are referred to as α:β (or αβ) T cells, though a minority of T cells express an alternate receptor, formed by variable gamma (γ) and delta (δ) chains, referred asγδ T cells.[11]
Each chain is composed of two extracellular domains: Variable (V) region and a Constant (C) region, both ofImmunoglobulin superfamily(IgSF)domainforming antiparallelβ-sheets.The Constant region is proximal to the cell membrane, followed by a transmembrane region and a short cytoplasmic tail, while the Variable region binds to the peptide/MHC complex.
The variable domain of both the TCR α-chain and β-chain each have three hypervariable orcomplementarity-determining regions(CDRs). There is also an additional area of hypervariability on the β-chain (HV4) that does not normally contact antigen and, therefore, is not considered a CDR.[citation needed]
The residues in these variable domains are located in two regions of the TCR, at the interface of the α- and β-chains and in the β-chainframework regionthat is thought to be in proximity to the CD3 signal-transduction complex.[12]CDR3 is the main CDR responsible for recognizingprocessed antigen,although CDR1 of the alpha chain has also been shown to interact with theN-terminalpart of the antigenic peptide, whereas CDR1 of the β-chain interacts with theC-terminalpart of the peptide[citation needed].
CDR2 is thought to recognize the MHC. HV4 of the β-chain is not thought to participate in antigen recognition as in classical CDRs, but has been shown to interact withsuperantigens.[13]
The constant domain of the TCR consists of short connecting sequences in which a cysteine residue forms disulfide bonds, which form a link between the two chains.
The TCR is a member of the immunoglobulin superfamily, a large group of proteins involved in binding, recognition, and adhesion; the family is named afterantibodies(also called immunoglobulins). The TCR is similar to a half-antibody consisting of a single heavy and single light chain, except the heavy chain is without its crystallizable fraction (Fc). The two main subunits of TCR (α- and β-chains) are twisted together. CD3 and zeta subunits are required to carry out the signal transduction. The MHC-TCR-CD3 interaction for T cells is functionally similar to the antigen(Ag)-immunoglobulin(Ig)-FcR interaction for myeloid leukocytes, and Ag-Ig-CD79 interaction for B cells.
Generation of the TCR diversity
editThe generation of TCR diversity is similar to that forantibodiesandB-cell antigen receptors.It arises mainly fromgenetic recombinationof the DNA-encoded segments in individual somatic T cells bysomatic V(D)J recombinationusingRAG1andRAG2recombinases. Unlikeimmunoglobulins,however, TCR genes do not undergosomatic hypermutation,and T cells do not expressactivation-induced cytidine deaminase(AID). The recombination process that creates diversity inBCR(antibodies) and TCR is unique tolymphocytes(T and B cells) during the early stages of their development in primary lymphoid organs (thymusfor T cells,bone marrowfor B cells).
Each recombined TCR possess uniqueantigenspecificity, determined by the structure of theantigen-binding siteformed by the α and β chains in case of αβ T cells or γ and δ chains on case of γδ T cells.[14]
- The TCRalpha chainis generated byVJ recombination,whereas thebeta chainis generated by VDJ recombination (both involving a random joining of gene segments to generate the complete TCR chain).
- Likewise, generation of the TCRgamma chaininvolves VJ recombination, whereas generation of the TCRdelta chainoccurs by VDJ recombination.
The intersection of these specific regions (V and J for the alpha or gamma chain; V, D, and J for the beta or delta chain) corresponds to the CDR3 region that is important for peptide/MHC recognition (see above).
It is the unique combination of the segments at this region, along withpalindromicand random nucleotide additions (respectively termed "P-" and "N-" ), which accounts for the even greater diversity of T-cell receptor specificity for processed antigenic peptides.
Later during development, individualCDR loopsof TCR can be re-edited in the periphery outside thymus by reactivation of recombinases using a process termedTCR revision(editing) and change its antigenic specificity.
The TCR complex
editIn the plasma membrane the TCR receptor chains α and β associate with six additional adaptor proteins to form an octameric complex. The complex contains both α and β chains, forming the ligand-binding site, and the signaling modulesCD3δ, CD3γ, CD3ε andCD3ζin the stoichiometry TCR α β - CD3εγ - CD3εδ - CD3ζζ. Charged residues in the transmembrane domain of each subunit form polar interactions allowing a correct and stable assembly of the complex.[15]Thecytoplasmictail of the TCR is very short, hence the CD3 adaptor proteins containing the signaling motifs are needed for propagating the signal from the triggered TCR into the cell.
The signaling motifs involved in TCR signaling are tyrosine residues in the cytoplasmic tail of these adaptor proteins that can be phosphorylated in the event of TCR-pMHC binding. The tyrosine residues reside in a specific amino acid sequence of the signature Yxx(L/I)x6-8Yxx(L/I), where Y, L, I indicate tyrosine, leucine and isoleucine residues, x denotes any amino acids, the subscript 6-8 indicates a sequence of 6 to 8 amino acids in length. This motif is very common in activator receptors of thenon-catalytic tyrosine-phosphorylated receptor(NTR) family and is referred to asimmunoreceptor tyrosine-based activation motif(ITAM).[5]CD3δ, CD3γ and CD3ε each contain a single ITAM, while CD3ζ contains three ITAMs. In total the TCR complex contains 10 ITAMs.[15]Phosphorylated ITAMs act as binding site for SH2-domains of additionally recruited proteins.
Antigen discrimination
editEach T cell expresses clonal TCRs which recognize a specific peptide loaded on aMHCmolecule (pMHC), either onMHC class IIon the surface ofantigen-presenting cellsorMHC class Ion any other cell type.[16] A unique feature of T cells is their ability to discriminate between peptides derived from healthy, endogenous cells and peptides from foreign or abnormal (e.g. infected or cancerous) cells in the body.[17]Antigen-presenting cells do not discriminate between self and foreign peptides and typically express a large number of self-derived pMHCs on their cell surface and only a few copies of any foreign pMHC. For example, cells infected with HIV have only 8–46 HIV-specific pMHCs, compared with 100,000 total pMHCs, per cell.[18][19]
Because T cells undergo positive selection in the thymus, there is a non-negligible affinity between self-pMHC and the TCR. Nevertheless, the T-cell receptor signaling should not be activated by self-pMHC so that endogenous, healthy cells are ignored by T cells. However, when these very same cells contain even minute quantities of pathogen-derived pMHC, T cells must get activated and initiate immune responses. The ability of T cells to ignore healthy cells but respond when these same cells express a small number of foreign pMHCs is known as antigen discrimination.[20][21]
To do so, T cells have a very high degree of antigen specificity, despite the fact that the affinity to the peptide/MHC ligand is rather low in comparison to other receptor types.[22]The affinity, given as thedissociation constant(Kd), between a TCR and a pMHC was determined bysurface plasmon resonance(SPR) to be in the range of 1–100 μM, with an association rate (kon) of 1000 -10000 M−1s−1and a dissociation rate (koff) of 0.01 -0.1 s−1.[23]In comparison, cytokines have an affinity of KD = 10–600 pM to their receptor.[24] It has been shown that even a single amino acid change in the presented peptide that affects the affinity of the pMHC to the TCR reduces the T-cell response and cannot be compensated by a higher pMHC concentration.[25]A negative correlation between the dissociation rate of the pMHC-TCR complex and the strength of the T-cell response has been observed.[26]That means, pMHC that bind the TCR for a longer time initiate a stronger activation of the T cell. Furthermore, T cells are highly sensitive; interaction with a single pMHC is enough to trigger activation.[27]T cells move on quickly from antigens that do not trigger responses, rapidly scanning pMHC on an antigen-presenting cell (APC) to increase the chance of finding a specific pMHC. On average, a T cell encounters 20 APCs per hour.[28]
Different models for the molecular mechanisms that underlie this highly specific and highly sensitive process of antigen discrimination have been proposed. The occupational model simply suggests that the TCR response is proportional to the number of pMHC bound to the receptor. Given this model, a shorter lifetime of a peptide can be compensated by higher concentration such that the maximum response of the T cell stays the same. However, this cannot be seen in experiments and the model has been widely rejected.[26] The most accepted view is that the TCR engages in kinetic proofreading. Thekinetic proofreadingmodel proposes that a signal is not directly produced upon binding but a series of intermediate steps ensure a time delay between binding and signal output. Such intermediate "proofreading" steps can be multiple rounds of tyrosine phosphorylation. These steps require energy and therefore do not happen spontaneously, only when the receptor is bound to its ligand. This way only ligands with high affinity that bind the TCR for a long enough time can initiate a signal. All intermediate steps are reversible, such that upon ligand dissociation the receptor reverts to its original unphosphorylated state before a new ligand binds.[29] This model predicts that maximum response of T cells decreases for pMHC with shorter lifetime. Experiments have confirmed this model.[26] However, the basic kinetic proofreading model has a trade-off between sensitivity and specificity. Increasing the number of proofreading steps increases the specificity but lowers the sensitivity of the receptor. The model is therefore not sufficient to explain the high sensitivity and specificity of TCRs that have been observed. (Altan Bonnet2005) Multiple models that extend the kinetic proofreading model have been proposed, but evidence for the models is still controversial.[17][30][31]
The antigen sensitivity is higher in antigen-experienced T cells than in naive T cells. Naive T cells pass through the process of functional avidity maturation with no change in affinity. It is based on the fact that effector and memory (antigen-experienced) T cell are less dependent on costimulatory signals and higher antigen concentration than naive T cell.[32]
Signaling pathway
editThe essential function of the TCR complex is to identify specific bound antigen derived from a potentially harmful pathogen and elicit a distinct and critical response. At the same time it has to ignore any self-antigen and tolerate harmless antigens such as food antigens. The signal transduction mechanism by which a T cell elicits this response upon contact with its unique antigen is termed T-cell activation. Upon binding to pMHC, the TCR initiates a signaling cascade, involving transcription factor activation and cytoskeletal remodeling resulting in T-cell activation. Active T cells secrete cytokines, undergo rapid proliferation, have cytotoxic activity and differentiate into effector and memory cells. When the TCR is triggered, T cells form an immunological synapse allowing them to stay in contact with theantigen presenting cellfor several hours.[33] On a population level, T-cell activation depends on the strength of TCR stimulation, thedose–response curveof ligand to cytokine production is sigmoidal. However, T-cell activation on a single cell level can be characterized by a digital switch-like response, meaning the T cell is fully activated if the stimulus is higher than a given threshold; otherwise the T cell stays in its non-activated state. There is no intermediate activation state. The robust sigmoid dose-response curve on population level results from individual T cells having slightly different thresholds.[25]
T cells need three signals to become fully activated. Signal 1 is provided by the T-cell receptor when recognising a specific antigen on a MHC molecule. Signal 2 comes fromco-stimulatory receptorson T cell such asCD28,triggered via ligands presented on the surface of other immune cells such as CD80 and CD86. These co-stimulatory receptors are expressed only when an infection or inflammatory stimulus is detected by the innate immune system, Known as a "Danger signal". This two-signal system makes sure that T cells only respond to harmful stimuli (i.e. pathogens or injury) and not to self-antigens. An additional third signal is provided bycytokines,which regulate the differentiation of T cells into different subsets of effector T cells.[33] There are a myriad of molecules involved in the complex biochemical process (calledtrans-membrane signaling) by which T-cell activation occurs. Below, the signaling cascade is described in detail.
Receptor activation
editThe initial triggering follows the mechanism common for allNTR receptor familymembers. Once the TCR binds a specific pMHC, the tyrosine residues of theimmunoreceptor tyrosine-based activation motifs(ITAMs) in itsCD3adaptor proteins are phosphorylated. The residues serve as docking sites for downstream signaling molecules, which can propagate the signal.[34][35] Phosphorylation of ITAMs is mediated by theSrc kinaseLck.Lck is anchored to the plasma membrane by associating with theco-receptorCD4orCD8,depending on the T-cell subtype. CD4 is expressed onhelper T cellsandregulatory T cells,and is specific forMHC class II.CD8, on the other hand, specific forMHC class I,is expressed oncytotoxic T cells. Binding of the co-receptor to the MHC brings Lck in close proximity to the CD3 ITAMs. It has been shown that 40% of Lck is active even before the TCR binds pMHC and therefore has the ability to constantly phosphorylate the TCR.[36]Tonic TCR signalling is avoided by the presence ofphosphataseCD45that removes phosphorylation from tyrosine residues and inhibits signal initiation. Upon binding the balance of kinase activity to phosphatase activity is perturbed, leading to a surplus of phosphorylation and initiation of the signal. How such perturbation is accomplished by TCR binding is still debated.Mechanismsinvolving conformational change of TCR, TCR aggregation andkinetic segregationhave been suggested.[34] Tyrosine kinaseFynmight be involved in ITAM phosphorylation but is not essential for TCR signaling.[37][38]
Proximal TCR signaling
editPhosphorylatedITAMsin the cytoplasmic tails of CD3 recruit protein tyrosine kinaseZap70that can bind to the phosphorylated tyrosine residues with itsSH2 domain.This brings Zap70 into close proximity to Lck which results to its phosphorylation and activation by Lck.[39]Lck phosphorylates a number of different proteins in the TCR pathway.[40] Once activated, Zap70 is able to phosphorylate multiple tyrosine residues of the transmembrane proteinLAT. LAT is ascaffold proteinassociated with the membrane. It itself does not have any catalytic activity but it provides binding sites for signalling molecules via phosphorylated tyrosine residues. LAT associates with another scaffolding proteinSlp-76via theGrap2adaptor protein, which provides additional binding sites. Together LAT and Slp-76 provide a platform for the recruitment of many downstream signaling molecules. By bringing these signalling molecules into close proximity, they can then be activated by Lck, Zap70 and other kinases. Therefore, the LAT/Slp76 complex act as a highly cooperative signalosome.[39]
Molecules that bind the LAT/Slp76 complex include:Phospholipase Cγ1 (PLCγ1),SOSvia aGrb2adaptor,Itk,Vav,Nck1andFyb.[39]
Signal transduction to the nucleus
editPLCγis a very important enzyme in the pathway as it generatessecond messengermolecules. It is activated by the tyrosine kinase Itk which is recruited to the cell membrane by binding tophosphatidylinositol (3,4,5)-trisphosphate(PIP3). PIP3 is produced by the action ofphosphoinositide 3-kinase(PI-3K), which phosphorylatesphosphatidylinositol 4,5-bisphosphate(PIP2) to produce PIP3. It is not known that PI-3K is activated by the T-cell receptor itself, but there is evidence that CD28, a co-stimulatory receptor providing the second signal, is able to activate PI-3K. The interaction between PLCγ, Itk and PI-3K could be the point in the pathway where the first and the second signal are integrated. Only if both signals are present, PLCγ is activated.[33] Once PLCγ is activated by phosphorylation. It hydrolyses PIP2 into twosecondary messengermolecules, namely the membrane-bounddiacyl glycerol(DAG) and the solubleinositol 1,4,5-trisphosphate(IP3).[41]
These second messenger molecules amplify the TCR signal and distribute the prior localized activation to the entire cell and activate protein cascades that finally lead to the activation oftranscription factors. Transcription factors involved in T-cell signaling pathway are theNFAT,NF-κBandAP1,aheterodimerof proteinsFosandJun.All three transcription factors are needed to activate the transcription ofinterleukin-2(IL2) gene.[33]
NFAT
editNFATactivation depends oncalcium signaling. IP3 produced by PLC-γ is no longer bound to the membrane and diffuses rapidly in the cell. Binding of IP3 tocalcium channel receptorson theendoplasmic reticulum(ER) induces the release of calcium (Ca2+) into the cytosol. The resulting low Ca2+concentration in the ER causesSTIM1clustering on the ER membrane, which in turn leads to activation of cell membraneCRACchannels that allows additional calcium to flow into the cytosol from the extracellular space. Therefore, levels of Ca2+are strongly increased in the T cell. This cytosolic calcium bindscalmodulin,inducing a conformational change of the protein such that it can then bind and activatecalcineurin.Calcineurin, in turn, dephosphorylates NFAT. In its deactivated state, NFAT cannot enter thenucleusas itsnuclear localization sequence(NLS) cannot be recognized by nuclear transporters due to phosphorylation byGSK-3.When dephosphorylated by Calcineurin translocation of NFAT into the nucleus is possible.[33] Additionally, there is evidence that PI-3K via signal molecules recruits the protein kinaseAKTto the cell membrane. AKT is able to deactivate GSK3 and thereby inhibiting the phosphorylation of NFAT, which could contribute to NFAT activation.[39]
NF-κB
editNF-κBactivation is initiated by DAG, the second, membrane bound product of PLCγ hydrolyzation of PIP2. DAG binds and recruitsprotein kinase Cθ (PKCθ) to the membrane where it can activate the membrane bound scaffold proteinCARMA1.CARMA1 then undergoes a conformational change which allows it to oligomerize and bind the adapter proteinsBCL10,CARD domainandMALT1.This multi-subunit complex binds theubiquitin ligaseTRAF6.Ubiquitinationof TRAF6 serves as scaffold to recruitNEMO,IκB kinase(IKK) andTAK1.[33]TAK 1 phosphorylates IKK, which in turn phosphorylates the NF-κB inhibitorI-κB,leading to the ubiquitination and subsequent degradation of I-κB. I-κB blocks the NLS of NF-κB therefore preventing its translocation to the nucleus. Once I-κB is degraded, it cannot bind to NF-κB and the NLS of NF-κB becomes accessible for nuclear translocation.[33]
AP1
editActivation ofAP1 factorinvolves threeMAPK signaling pathways.These pathways use a phosphorylation cascade of three successive acting protein kinases to transmit a signal. The three MAPK pathways in T cells involve kinases of different specificities belonging to each of theMAP3K,MAP2K,MAPKfamilies. Initial activation is done by theGTPaseRasorRacwhich phosphorylate the MAP3K.[33] A cascade involving the enzymesRaf,MEK1,ERKresults in the phosphorylation of Jun, conformational change allows Jun to bind to Fos and hence AP-1 to form. AP-1 then acts as transcription factor. Raf is activated via the second messenger DAG, SOS, and Ras. DAG recruits among other proteins the RAS guanyl nucleotide-releasing protein (RasGRP), aguanine nucleotide exchange factor(GEF), to the membrane. RasGRP activates the small GTPase Ras by exchangingguanosine diphosphate(GDP) bound to Ras againstguanosine triphosphate(GTP). Ras can also be activated by the guanine nucleotide exchange factor SOS which binds to the LAT signalosome. Ras then initiates the MAPK cascade.[39] The second MAPK cascade withMEKK1,JNKK,JNKinduces protein expression of Jun. Another cascade, also involving MEKK1 as MAPK3, but then activatingMKK3/6andp38induces Fos transcription. Activation of MEKK1, additionally to being activated by Ras, involves Slp-76 recruiting the GEF Vav to the LAT signalosome, which then activates the GTPase Rac. Rac and Ras activate MEKK1 and thereby initiate the MAPK cascade.[39]
See also
editReferences
edit- ^Kindt TJ, Goldsby RA, Osborne BA, Kuby J (2007).Kuby immunology.Macmillan. pp. 223–.ISBN978-1-4292-0211-4.Retrieved28 November2010.
- ^Sewell AK (September 2012)."Why must T cells be cross-reactive?".Nature Reviews. Immunology.12(9):669–77.doi:10.1038/nri3279.PMC7097784.PMID22918468.
- ^abGlusman G, Rowen L, Lee I, Boysen C, Roach JC, Smit AF, et al. (September 2001)."Comparative genomics of the human and mouse T cell receptor loci".Immunity.15(3):337–49.doi:10.1016/s1074-7613(01)00200-x.PMID11567625.
- ^Deakin JE, Parra ZE, Graves JA, Miller RD (2006)."Physical mapping of T cell receptor loci (TRA@, TRB@, TRD@ and TRG@) in the opossum (Monodelphis domestica)".Cytogenetic and Genome Research.112(3–4): 342K.doi:10.1159/000089901.PMID16484802.
- ^abDushek O, Goyette J, van der Merwe PA (November 2012). "Non-catalytic tyrosine-phosphorylated receptors".Immunological Reviews.250(1):258–76.doi:10.1111/imr.12008.PMID23046135.S2CID1549902.
- ^Allison JP, McIntyre BW, Bloch D (November 1982)."Tumor-specific antigen of murine T-lymphoma defined with monoclonal antibody".Journal of Immunology.129(5):2293–2300.doi:10.4049/jimmunol.129.5.2293.PMID6181166.S2CID13249566.
- ^Meuer SC, Fitzgerald KA, Hussey RE, Hodgdon JC, Schlossman SF, Reinherz EL (February 1983)."Clonotypic structures involved in antigen-specific human T cell function. Relationship to the T3 molecular complex".The Journal of Experimental Medicine.157(2):705–719.doi:10.1084/jem.157.2.705.PMC2186929.PMID6185617.
- ^Haskins K, Kubo R, White J, Pigeon M, Kappler J, Marrack P (April 1983)."The major histocompatibility complex-restricted antigen receptor on T cells. I. Isolation with a monoclonal antibody".The Journal of Experimental Medicine.157(4):1149–1169.doi:10.1084/jem.157.4.1149.PMC2186983.PMID6601175.
- ^Yanagi Y, Yoshikai Y, Leggett K, Clark SP, Aleksander I, Mak TW (8 March 1984). "A human T cell-specific cDNA clone encodes a protein having extensive homology to immunoglobulin chains".Nature.308(5955):145–149.Bibcode:1984Natur.308..145Y.doi:10.1038/308145a0.PMID6336315.S2CID4229210.
- ^Hedrick SM, Cohen DI, Nielsen EA, Davis MM (8 March 1984). "Isolation of cDNA clones encoding T cell-specific membrane-associated proteins".Nature.308(5955):149–153.Bibcode:1984Natur.308..149H.doi:10.1038/308149a0.PMID6199676.S2CID4273688.
- ^Janeway Jr CA, Travers P, Walport M, et al. (2001).Immunobiology: The Immune System in Health and Disease. 5th edition.Glossary: Garland Science.
- ^Kieke MC, Shusta EV, Boder ET, Teyton L, Wittrup KD, Kranz DM (May 1999)."Selection of functional T cell receptor mutants from a yeast surface-display library".Proceedings of the National Academy of Sciences of the United States of America.96(10):5651–6.Bibcode:1999PNAS...96.5651K.doi:10.1073/pnas.96.10.5651.PMC21915.PMID10318939.
- ^Sundberg EJ, Deng L, Mariuzza RA (August 2007)."TCR recognition of peptide/MHC class II complexes and superantigens".Seminars in Immunology.The Structure and Function of Antigen Receptors.19(4):262–271.doi:10.1016/j.smim.2007.04.006.PMC2949352.PMID17560120.
- ^Janeway CA, Travers P, Walport M, et al. (2001)."The Generation of Lymphocyte Antigen Receptors".Immunobiology: The Immune System in Health and Disease(5th ed.). Garland Science.
- ^abCall ME, Pyrdol J, Wiedmann M, Wucherpfennig KW (December 2002)."The organizing principle in the formation of the T cell receptor-CD3 complex".Cell.111(7):967–79.doi:10.1016/s0092-8674(02)01194-7.PMC3420808.PMID12507424.
- ^Smith-Garvin JE, Koretzky GA, Jordan MS (2009)."T cell activation".Annual Review of Immunology.27:591–619.doi:10.1146/annurev.immunol.021908.132706.PMC2740335.PMID19132916.
- ^abFeinerman O, Germain RN, Altan-Bonnet G (February 2008)."Quantitative challenges in understanding ligand discrimination by alphabeta T cells".Molecular Immunology.45(3):619–31.doi:10.1016/j.molimm.2007.03.028.PMC2131735.PMID17825415.
- ^Yang H, Buisson S, Bossi G, Wallace Z, Hancock G, So C, et al. (November 2016)."Elimination of Latently HIV-infected Cells from Antiretroviral Therapy-suppressed Subjects by Engineered Immune-mobilizing T-cell Receptors".Molecular Therapy.24(11):1913–1925.doi:10.1038/mt.2016.114.PMC5154472.PMID27401039.
- ^Blum JS, Wearsch PA, Cresswell P (2013)."Pathways of antigen processing".Annual Review of Immunology.31:443–73.doi:10.1146/annurev-immunol-032712-095910.PMC4026165.PMID23298205.
- ^Evavold BD, Allen PM (May 1991). "Separation of IL-4 production from Th cell proliferation by an altered T cell receptor ligand".Science.252(5010):1308–10.Bibcode:1991Sci...252.1308E.doi:10.1126/science.1833816.PMID1833816.
- ^Kersh GJ, Allen PM (October 1996)."Structural basis for T cell recognition of altered peptide ligands: a single T cell receptor can productively recognize a large continuum of related ligands".The Journal of Experimental Medicine.184(4):1259–68.doi:10.1084/jem.184.4.1259.PMC2192852.PMID8879197.
- ^Donermeyer DL, Weber KS, Kranz DM, Allen PM (November 2006)."The study of high-affinity TCRs reveals duality in T cell recognition of antigen: specificity and degeneracy".Journal of Immunology.177(10):6911–9.doi:10.4049/jimmunol.177.10.6911.PMID17082606.
- ^Cole DK, Pumphrey NJ, Boulter JM, Sami M, Bell JI, Gostick E, et al. (May 2007)."Human TCR-binding affinity is governed by MHC class restriction".Journal of Immunology.178(9):5727–34.doi:10.4049/jimmunol.178.9.5727.PMID17442956.
- ^Whitty A, Raskin N, Olson DL, Borysenko CW, Ambrose CM, Benjamin CD, Burkly LC (October 1998)."Interaction affinity between cytokine receptor components on the cell surface".Proceedings of the National Academy of Sciences of the United States of America.95(22):13165–70.Bibcode:1998PNAS...9513165W.doi:10.1073/pnas.95.22.13165.PMC23746.PMID9789059.
- ^abAltan-Bonnet G, Germain RN (November 2005)."Modeling T cell antigen discrimination based on feedback control of digital ERK responses".PLOS Biology.3(11): e356.doi:10.1371/journal.pbio.0030356.PMC1262625.PMID16231973.
- ^abcDushek O, Aleksic M, Wheeler RJ, Zhang H, Cordoba SP, Peng YC, et al. (June 2011)."Antigen potency and maximal efficacy reveal a mechanism of efficient T cell activation".Science Signaling.4(176): ra39.doi:10.1126/scisignal.2001430.PMC4143974.PMID21653229.
- ^Huang J, Brameshuber M, Zeng X, Xie J, Li QJ, Chien YH, et al. (November 2013)."A single peptide-major histocompatibility complex ligand triggers digital cytokine secretion in CD4(+) T cells".Immunity.39(5):846–57.doi:10.1016/j.immuni.2013.08.036.PMC3846396.PMID24120362.
- ^Miller MJ, Hejazi AS, Wei SH, Cahalan MD, Parker I (January 2004)."T cell repertoire scanning is promoted by dynamic dendritic cell behavior and random T cell motility in the lymph node".Proceedings of the National Academy of Sciences of the United States of America.101(4):998–1003.Bibcode:2004PNAS..101..998M.doi:10.1073/pnas.0306407101.PMC327133.PMID14722354.
- ^McKeithan TW (May 1995)."Kinetic proofreading in T-cell receptor signal transduction".Proceedings of the National Academy of Sciences of the United States of America.92(11):5042–6.Bibcode:1995PNAS...92.5042M.doi:10.1073/pnas.92.11.5042.PMC41844.PMID7761445.
- ^Dushek O, van der Merwe PA (April 2014)."An induced rebinding model of antigen discrimination".Trends in Immunology.35(4):153–8.doi:10.1016/j.it.2014.02.002.PMC3989030.PMID24636916.
- ^Lever M, Maini PK, van der Merwe PA, Dushek O (September 2014)."Phenotypic models of T cell activation".Nature Reviews. Immunology.14(9):619–29.doi:10.1038/nri3728.PMID25145757.S2CID14274400.
- ^von Essen MR, Kongsbak M, Geisler C (2012)."Mechanisms behind functional avidity maturation in T cells".Clinical & Developmental Immunology.2012:163453.doi:10.1155/2012/163453.PMC3351025.PMID22611418.
- ^abcdefghMurphy KM, Weaver C (22 March 2016).Janeway's immunobiology(Ninth ed.). W.W. Norton & Company.ISBN978-0815345510.
- ^abvan der Merwe PA, Dushek O (2011). "Mechanisms for T cell receptor triggering".Nature Reviews Immunology.11(1):47–55.doi:10.1038/nri2887.PMID21127503.S2CID22423010.
- ^Abram CL, Lowell CA (March 2007). "The expanding role for ITAM-based signaling pathways in immune cells".Science's STKE.2007(377): re2.doi:10.1126/stke.3772007re2.PMID17356173.S2CID44314604.
- ^Nika K, Soldani C, Salek M, Paster W, Gray A, Etzensperger R, et al. (June 2010)."Constitutively active Lck kinase in T cells drives antigen receptor signal transduction".Immunity.32(6):766–77.doi:10.1016/j.immuni.2010.05.011.PMC2996607.PMID20541955.
- ^Tang Q, Subudhi SK, Henriksen KJ, Long CG, Vives F, Bluestone JA (May 2002)."The Src family kinase Fyn mediates signals induced by TCR antagonists".Journal of Immunology.168(9):4480–7.doi:10.4049/jimmunol.168.9.4480.PMID11970992.
- ^Salmond RJ, Filby A, Qureshi I, Caserta S, Zamoyska R (March 2009). "T-cell receptor proximal signaling via the Src-family kinases, Lck and Fyn, influences T-cell activation, differentiation, and tolerance".Immunological Reviews.228(1):9–22.doi:10.1111/j.1600-065X.2008.00745.x.PMID19290918.S2CID46343285.
- ^abcdefHuse M (May 2009)."The T-cell-receptor signaling network".Journal of Cell Science.122(Pt 9):1269–73.doi:10.1242/jcs.042762.PMID19386893.
- ^"UniProtKB - P06239 (LCK_HUMAN)".Uniprot.Retrieved7 May2020.
- ^Essen LO, Perisic O, Katan M, Wu Y, Roberts MF, Williams RL (February 1997). "Structural mapping of the catalytic mechanism for a mammalian phosphoinositide-specific phospholipase C".Biochemistry.36(7):1704–18.doi:10.1021/bi962512p.PMID9048554.
External links
edit- T-Cell+Receptorat the U.S. National Library of MedicineMedical Subject Headings(MeSH)