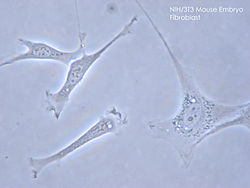
Afibroblastis a type ofbiological celltypically with a spindle shape[1]that synthesizes theextracellular matrixandcollagen,[2]produces the structural framework (stroma) for animaltissues,and plays a critical role inwound healing.[3]Fibroblasts are the most common cells ofconnective tissuein animals.
| Fibroblast | |
|---|---|
 | |
| Details | |
| Location | Connective tissue |
| Function | Extracellular matrixandcollagencreation |
| Identifiers | |
| Latin | fibroblastus |
| MeSH | D005347 |
| TH | H2.00.03.0.01002 |
| FMA | 63877 |
| Anatomical terms of microanatomy | |
Structure
editThis sectionneeds additional citations forverification.(April 2021) |
Fibroblasts have a branchedcytoplasmsurrounding an elliptical, specklednucleushaving two or morenucleoli.Active fibroblasts can be recognized by their abundantrough endoplasmic reticulum(RER). Inactive fibroblasts, called 'fibrocytes', are smaller, spindle-shaped, and have less RER. Although disjointed and scattered when covering large spaces, fibroblasts often locally align in parallel clusters when crowded together.
Unlike theepithelialcells lining the body structures, fibroblasts do not form flat monolayers and are not restricted by a polarizing attachment to abasal laminaon one side, although they may contribute to basal lamina components in some situations (e.g. subepithelialmyofibroblastsinintestinemay secrete the α-2 chain-carrying component of thelaminin,which is absent only in regions of follicle-associated epithelia which lack the myofibroblast lining). Fibroblasts can also migrate slowly over substratum as individual cells, again in contrast to epithelial cells. While epithelial cells form the lining of body structures, fibroblasts and related connective tissues sculpt the "bulk" of an organism.
The life span of a fibroblast, as measured inchickembryos, is 57 ± 3 days.[4]
Relationship with fibrocytes
editFibroblasts and fibrocytes are two states of the same cells, the former being the activated state, the latter the less active state, concerned with maintenance and tissue metabolism. Currently, there is a tendency to call both forms fibroblasts. The suffix "-blast" is used in cellular biology to denote astem cellor a cell in an activated state ofmetabolism.
Fibroblasts are morphologically heterogeneous with diverse appearances depending on their location and activity. Though morphologically inconspicuous,ectopicallytransplanted fibroblasts can often retainpositional memoryof the location and tissue context where they had previously resided, at least over a few generations.[5]This remarkable behavior may lead to discomfort[clarification needed]in the rare event that they stagnate there excessively.
Development
editThe main function of fibroblasts is to maintain the structural integrity of connective tissues by continuously secreting precursors of theextracellular matrix(ECM), providing all such components, primarily theground substanceand a variety offibers.The composition of the ECM determines the physical properties of connective tissues.
Like other cells of connective tissue, fibroblasts are derived from primitivemesenchyme.Hence, they express the intermediate filament proteinvimentin,a feature used as a marker to distinguish theirmesodermalorigin.[6]However, this test is not specific as epithelial cells cultured in vitro on adherent substratum may also express vimentin after some time.
In certain situations, epithelial cells can give rise to fibroblasts, a process calledepithelial-mesenchymal transition.
Conversely, fibroblasts in some situations may give rise to epithelia by undergoing a mesenchymal to epithelial transition and organizing into a condensed, polarized, laterally connected true epithelial sheet. This process is seen in many developmental situations (e.g.nephronandnotocorddevelopment), as well as in wound healing and tumorigenesis.[citation needed]
Function
editFibroblasts makecollagenfibers,glycosaminoglycans,reticularandelastic fibers.The fibroblasts of growing individuals divide and synthesize ground substance. Tissue damage stimulates fibrocytes and induces the production of fibroblasts.[7]
Inflammation
editBesides their commonly known role as structural components, fibroblasts play a critical role in an immune response to a tissue injury. They are early players in initiating inflammation in the presence of invading microorganisms. They induce chemokine synthesis through the presentation of receptors on their surface. Immune cells then respond and initiate a cascade of events to clear the invasive microorganisms. Receptors on the surface of fibroblasts also allow regulation ofhematopoietic cellsand provide a pathway for immune cells to regulate fibroblasts.[8]
Tumour mediation
editFibroblasts, liketumor-associated host fibroblasts(TAF), play a crucial role in immune regulation through TAF-derived ECM components and modulators. TAF are known to be significant in the inflammatory response as well as immune suppression in tumors. TAF-derived ECM components cause alterations in ECM composition and initiate the ECM remodeling.[9]ECM remodeling is described as changes in the ECM as a result ofenzymeactivity which can lead to degradation of the ECM. Immune regulation of tumors is largely determined by ECM remodeling because the ECM is responsible for regulating a variety of functions, such as proliferation, differentiation, and morphogenesis of vital organs.[10]In many tumor types, especially those related to the epithelial cells, ECM remodeling is common. Examples of TAF-derived ECM components include Tenascin and Thrombospondin-1 (TSP-1), which can be found in sites of chronic inflammation and carcinomas, respectively.[9]
Immune regulation of tumors can also occur through the TAF-derived modulators. Although these modulators may sound similar to the TAF-derived ECM components, they differ in the sense that they are responsible for the variation and turnover of the ECM. Cleaved ECM molecules can play a critical role in immune regulation. Proteases like matrixmetalloproteineasesand the uPA system are known to cleave the ECM. These proteases are derived from fibroblasts.[9]
Use of fibroblasts as feeder cells
editMouse embryonic fibroblasts(MEFs) are often used as supportive "feeder cells" in research using human embryonic stem cells,[11]induced pluripotent stem cells and primary epithelial cell culture.[12]However, many researchers are trying to phase out MEFs in favor of culture media with precisely defined ingredients in order to facilitate the development of clinical-grade products.[13]
In view of the potential clinical applications of stem cell-derived tissues or primary epithelial cells, the use of human fibroblasts as an alternative to MEF feeders has been studied.[14]Whereas the fibroblasts are usually used to maintain pluripotency of the stem cells, they can also be used to facilitate development of the stem cells into specific type of cells such as cardiomyocytes.[15]
Host immune response
editFibroblasts from different anatomical sites in the body express many genes that code forimmune mediatorsand proteins.[16]These mediators of immune response enable thecellular communicationwith hematopoietic immune cells.[17]The immune activity of non-hematopoietic cells, such as fibroblasts, is referred to as “structural immunity”.[16][18]In order to facilitate a fast response to immunological challenges, fibroblasts encode crucial aspects of the structural cell immune response in theepigenome.[citation needed]
See also
editReferences
edit- ^Murray, Lynne A.; Knight, Darryl A.; Laurent, Geoffrey J. (2009). "Fibroblasts".Asthma and COPD:193–200.doi:10.1016/B978-0-12-374001-4.00015-8.
- ^"Fibroblast".Genetics Home Reference.U.S. National Library of Medicine. 2014-05-05.Retrieved2014-05-10.
- ^"Fibroblasts".Retrieved16 August2018.
- ^Weissman-Shomer P, Fry M (1975). "Chick embryo fibroblasts senscence in vitro: pattern of cell division and life span as a function of cell density".Mechanisms of Ageing and Development.4(2): 159–166.doi:10.1016/0047-6374(75)90017-2.PMID1152547.S2CID9299977.
- ^Advances in Extracellular Space Research and Application.Scholarly Editions. 2013. p. 251.ISBN9781481682626.
- ^Dave JM, Bayless KJ (May 2014). "Vimentin as an integral regulator of cell adhesion and endothelial sprouting".Microcirculation.21(4): 333–344.doi:10.1111/micc.12111.PMID24387004.S2CID26292524.
- ^Pilling, Darrell; Vakil, Varsha; Cox, Nehemiah; Gomer, Richard H. (2015-09-22)."TNF-α–stimulated fibroblasts secrete lumican to promote fibrocyte differentiation".Proceedings of the National Academy of Sciences.112(38): 11929–11934.doi:10.1073/pnas.1507387112.ISSN0027-8424.PMC4586854.PMID26351669.
- ^Smith RS, Smith TJ, Blieden TM, Phipps RP (August 1997)."Fibroblasts as sentinel cells. Synthesis of chemokines and regulation of inflammation".The American Journal of Pathology.151(2): 317–322.PMC1858004.PMID9250144.
- ^abcSilzle T, Randolph GJ, Kreutz M, Kunz-Schughart LA (January 2004). "The fibroblast: sentinel cell and local immune modulator in tumor tissue".International Journal of Cancer.108(2): 173–180.doi:10.1002/ijc.11542.PMID14639599.S2CID10936034.
- ^Bonnans C, Chou J, Werb Z (December 2014)."Remodelling the extracellular matrix in development and disease".Nature Reviews. Molecular Cell Biology.15(12): 786–801.doi:10.1038/nrm3904.PMC4316204.PMID25415508.
- ^Llames, S.; García-Pérez, E.; Meana, A.; Larcher, F.; del Río, M. (2015)."Feeder Layer Cell Actions and Applications".Tissue Eng Part B Rev.21(4): 345–353.doi:10.1089/ten.teb.2014.0547.PMC4533020.PMID25659081.
- ^Hynds, R.E.; Bonfanti, P.; Janes, S.M. (2018)."Regenerating human epithelia with cultured stem cells: feeder cells, organoids and beyond".EMBO Molecular Medicine.10(2): 139–150.doi:10.15252/emmm.201708213.PMC5801505.PMID29288165.
- ^Hagbard, L.; Cameron, K.; August, P.; Penton, C.; Parmar, M.; Hay, D.C.; Kallur, T. (2018)."Developing defined substrates for stem cell culture and differentiation".Philosophical Transactions of the Royal Society B.373(1750).doi:10.1098/rstb.2017.0230.PMC5974452.PMID29786564.
- ^Desai N, Rambhia P, Gishto A (February 2015)."Human embryonic stem cell cultivation: historical perspective and evolution of xeno-free culture systems".Reproductive Biology and Endocrinology.13(1): 9.doi:10.1186/s12958-015-0005-4.PMC4351689.PMID25890180.
- ^Matsuda Y, Takahashi K, Kamioka H, Naruse K (September 2018)."Human gingival fibroblast feeder cells promote maturation of induced pluripotent stem cells into cardiomyocytes".Biochemical and Biophysical Research Communications.503(3): 1798–1804.doi:10.1016/j.bbrc.2018.07.116.PMID30060947.
- ^abKrausgruber T, Fortelny N, Fife-Gernedl V, Senekowitsch M, Schuster LC, Lercher A, et al. (July 2020)."Structural cells are key regulators of organ-specific immune responses".Nature.583(7815): 296–302.Bibcode:2020Natur.583..296K.doi:10.1038/s41586-020-2424-4.PMC7610345.PMID32612232.S2CID220295181.
- ^Armingol E, Officer A, Harismendy O, Lewis NE (November 2020)."Deciphering cell-cell interactions and communication from gene expression".Nature Reviews. Genetics.22(2): 71–88.doi:10.1038/s41576-020-00292-x.PMC7649713.PMID33168968.
- ^Minton K (September 2020). "A gene atlas of 'structural immunity'".Nature Reviews. Immunology.20(9): 518–519.doi:10.1038/s41577-020-0398-y.PMID32661408.S2CID220491226.