Neurine
Appearance
| |
| Names | |
|---|---|
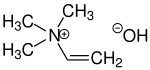
| IUPAC name
Trimethylvinylammonium hydroxide
| |
| Other names
Vitaloid;N,N,N-Trimethylethenaminium hydroxide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.006.678 |
PubChemCID
|
|
| UNII | |
CompTox Dashboard(EPA)
|
|
| |
| |
| Properties | |
| C5H13NO | |
| Molar mass | 103.16 |
| Appearance | Syrupy liquid |
| Soluble | |
Except where otherwise noted, data are given for materials in theirstandard state(at 25 °C [77 °F], 100 kPa).
| |
Neurineis analkaloidfound in egg yolk, brain, bile and in cadavers. It is formed during putrefaction of biological tissues by thedehydrationofcholine.It is a poisonous, syrupy liquid with a fishy odor.
Neurine is aquaternary ammonium saltwith threemethylgroups and onevinyl groupattached to the nitrogen atom. Synthetically, neurine can be prepared by the reaction ofacetylenewithtrimethylamine.[1]Neurine is unstable and decomposes readily to form trimethylamine.
References
[edit]- ^Gardner, C.; Kerrigan, V.; Rose, J. D.; Weedon, B. C. L. (1949-01-01)."169. Acetylene reactions. Part IV. Formation of trimethylvinyl- and tetramethyl-ammonium hydroxide from acetylene and aqueous trimethylamine".Journal of the Chemical Society (Resumed):789–792.doi:10.1039/JR9490000789.ISSN0368-1769.
- Merck Index,11th Edition,6393.
