3-Indolepropionic acid
This article needs to beupdated.The reason given is: recently published information on this compound from this[1]review.(June 2018) |
This articleis missing informationabout role in plants as a natural auxin; which plants make it and how farmers use it.(April 2022) |
 | |
| Clinical data | |
|---|---|
| Trade names | Oxigon[2] |
| Other names | Conjugate acid: • 1H-Indole-3-propanoic acid • Indole-3-propionic acid Conjugate base: •Indole-3-propionate |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChemCID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| CompTox Dashboard(EPA) | |
| ECHA InfoCard | 100.011.455 |
| Chemical and physical data | |
| Formula | C11H11NO2 |
| Molar mass | 189.214g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 134 to 135 °C (273 to 275 °F)[3] |
| |
| |
| (verify) | |
3-Indolepropionic acid(IPA), orindole-3-propionic acid,has been studied for its therapeutic value in the treatment ofAlzheimer's disease.As of 2022[4]IPA shows potential in the treatment of this disease, though the therapeutic effect of IPA depends on dose and time of therapy initiation.
Though promising in some historical clinical trials, IPA is not clinically listed as a useful therapeutic in managing Alzheimer's as of 2023.[5]
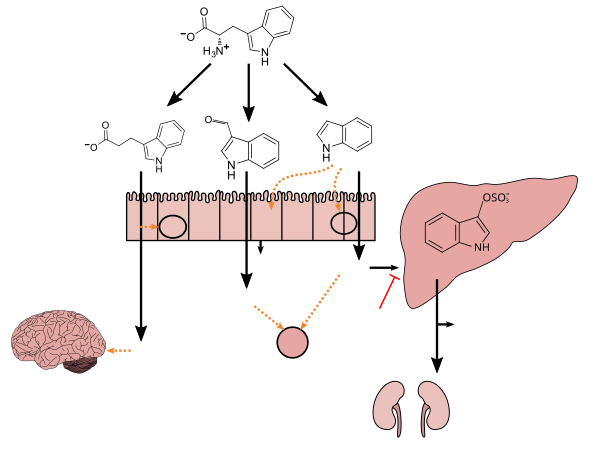
This compound endogenously produced byhuman microbiotaand has only been detectedin vivowhen the speciesClostridium sporogenesis present in the gastrointestinal tract.[6][7][8]As of April 2016[update],C. sporogenes,which usestryptophanto synthesize IPA, is the only species of bacteria known to synthesize IPAin vivoat levels which are subsequently detectable in theblood plasmaof the host.[6][7][8][9]
IPA is an even more potent scavenger ofhydroxyl radicalsthanmelatonin,the most potent scavenger of hydroxyl radicals that is synthesized by human enzymes.[3][9]Similar to melatonin but unlike other antioxidants, it scavenges radicals without subsequently generating reactive and pro-oxidant intermediate compounds.[3][9][10]In 2017, elevated concentrations of IPA in humanblood plasmawere found to be correlated with a lower risk oftype 2 diabetesand higher consumption offiber-rich foods.[3][11][12]
Biosynthesis in humans and cellular effects
[edit]Tryptophan metabolism byhuman gastrointestinal microbiota()
|
Metabolism
[edit]IPA can be converted in theliverorkidneysto3-indoleacrylic acid,which is subsequentlyconjugated with glycine,forming indolylacryloyl glycine.[13]
History
[edit]The neuroprotective, antioxidant, and anti-amyloid properties of IPA were first reported in 1999.[9][14][15][16]
See also
[edit]References
[edit]- ^Galligan JJ (February 2018). "Beneficial actions of microbiota-derived tryptophan metabolites".Neurogastroenterology and Motility.30(2): e13283.doi:10.1111/nmo.13283.PMID29341448.S2CID39904059.
- ^Bendheim PE, Poeggeler B, Neria E, Ziv V, Pappolla MA, Chain DG (October 2002). "Development of indole-3-propionic acid (OXIGON) for Alzheimer's disease".Journal of Molecular Neuroscience.19(1–2): 213–217.doi:10.1007/s12031-002-0036-0.PMID12212784.S2CID31107810.
The accumulation of amyloid-beta and concomitant oxidative stress are major pathogenic events in Alzheimer's disease. Indole-3-propionic acid (IPA, OXIGON) is a potent anti-oxidant devoid of pro-oxidant activity. IPA has been demonstrated to be an inhibitor of beta-amyloid fibril formation and to be a potent neuroprotectant against a variety of oxidotoxins. This review will summarize the known properties of IPA and outline the rationale behind its selection as a potential disease-modifying therapy for Alzheimer's disease.
- ^abcdef"3-Indolepropionic acid".Human Metabolome Database.University of Alberta.Retrieved12 June2018.
- ^Jiang H, Chen C, Gao J (December 2022)."Extensive Summary of the Important Roles of Indole Propionic Acid, a Gut Microbial Metabolite in Host Health and Disease".Nutrients.15(1): 151.doi:10.3390/nu15010151.PMC9824871.PMID36615808.
- ^"How Alzheimer's drugs help manage symptoms".Mayo Clinic.Retrieved3 November2023.
- ^abcWikoff WR, Anfora AT, Liu J, Schultz PG, Lesley SA, Peters EC,Siuzdak G(March 2009)."Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites".Proc. Natl. Acad. Sci. U.S.A.106(10): 3698–3703.Bibcode:2009PNAS..106.3698W.doi:10.1073/pnas.0812874106.PMC2656143.PMID19234110.
Production of IPA was shown to be completely dependent on the presence of gut microflora and could be established by colonization with the bacterium Clostridium sporogenes.... Conversely, a different set of enteric bacteria has been implicated in the metabolic transformation of indole to indole-3-propionic acid (IPA) (27). IPA, also identified only in the plasma of conv mice, has been shown to be a powerful antioxidant (28)... Although the presence of IPA in mammals has long been ascribed in the literature to bacterial metabolic processes, this conclusion was based on either the production of IPA in ex vivo cultures of individual bacterial species (31) or observed decreases in IPA levels in animals after administration of antibiotics (32). In our own survey of IPA production by representative members of the intestinal flora, only Clostridium sporogenes was found to produce IPA in culture (Table S2). Based on these results, individual GF mice were intentionally colonized with C. sporogenes strain ATCC 15579, and blood samples were taken at several intervals after colonization. IPA was undetectable in the samples taken shortly after introduction of the microbes, and was first observed in the serum 5 days after colonization, reaching plateau values comparable with conv mice by day 10. These colonization studies demonstrate that the introduction of enteric bacteria capable of IPA production in vivo into the gastrointestinal tract is sufficient to introduce IPA into the bloodstream of the host. Also, other GF animals were injected i.p. with either IPA (at 10, 20, or 40 mg/kg) or sterile PBS vehicle, and their serum concentrations of IPA were measured over time. As seen in Table S3, the high serum levels of IPA observed 1 h after injection decreased more than 90% within 5 h, showing that IPA is rapidly cleared from the blood, and that its presence in the serum of conv animals must result from continuous production from 1 or more bacterial species associated with the mammalian gut.
IPA metabolism diagram - ^abcdefghijkZhang LS, Davies SS (April 2016)."Microbial metabolism of dietary components to bioactive metabolites: opportunities for new therapeutic interventions".Genome Med.8(1): 46.doi:10.1186/s13073-016-0296-x.PMC4840492.PMID27102537.
Lactobacillusspp. convert tryptophan to indole-3-aldehyde (I3A) through unidentified enzymes [125].Clostridium sporogenesconvert tryptophan to IPA [6], likely via a tryptophan deaminase.... IPA also potently scavenges hydroxyl radicals
Table 2: Microbial metabolites: their synthesis, mechanisms of action, and effects on health and disease
Figure 1: Molecular mechanisms of action of indole and its metabolites on host physiology and disease - ^abAttwood G, Li D, Pacheco D, Tavendale M (June 2006)."Production of indolic compounds by rumen bacteria isolated from grazing ruminants".Journal of Applied Microbiology.100(6): 1261–1271.doi:10.1111/j.1365-2672.2006.02896.x.PMID16696673.S2CID35673610.
- ^abcdeChyan YJ, Poeggeler B, Omar RA, Chain DG, Frangione B, Ghiso J, Pappolla MA (July 1999)."Potent neuroprotective properties against the Alzheimer beta-amyloid by an endogenous melatonin-related indole structure, indole-3-propionic acid".J. Biol. Chem.274(31): 21937–21942.doi:10.1074/jbc.274.31.21937.PMID10419516.S2CID6630247.
[Indole-3-propionic acid (IPA)] has previously been identified in the plasma and cerebrospinal fluid of humans, but its functions are not known.... In kinetic competition experiments using free radical-trapping agents, the capacity of IPA to scavenge hydroxyl radicals exceeded that of melatonin, an indoleamine considered to be the most potent naturally occurring scavenger of free radicals. In contrast with other antioxidants, IPA was not converted to reactive intermediates with pro-oxidant activity.
- ^Reiter RJ, Guerrero JM, Garcia JJ, Acuña-Castroviejo D (November 1998). "Reactive oxygen intermediates, molecular damage, and aging. Relation to melatonin".Annals of the New York Academy of Sciences.854(1): 410–424.Bibcode:1998NYASA.854..410R.doi:10.1111/j.1749-6632.1998.tb09920.x.PMID9928448.S2CID29333394.
- ^de Mello VD, Paananen J, Lindström J, Lankinen MA, Shi L, Kuusisto J, et al. (April 2017)."Indolepropionic acid and novel lipid metabolites are associated with a lower risk of type 2 diabetes in the Finnish Diabetes Prevention Study".Scientific Reports.7:46337.Bibcode:2017NatSR...746337D.doi:10.1038/srep46337.PMC5387722.PMID28397877.
- ^Tuomainen M, Lindström J, Lehtonen M, Auriola S, Pihlajamäki J, Peltonen M, et al. (May 2018)."Associations of serum indolepropionic acid, a gut microbiota metabolite, with type 2 diabetes and low-grade inflammation in high-risk individuals".Nutrition & Diabetes.8(1): 35.doi:10.1038/s41387-018-0046-9.PMC5968030.PMID29795366.
- ^Keszthelyi D, Troost FJ, Masclee AA (December 2009). "Understanding the role of tryptophan and serotonin metabolism in gastrointestinal function".Neurogastroenterology and Motility.21(12): 1239–1249.doi:10.1111/j.1365-2982.2009.01370.x.PMID19650771.S2CID23568813.
Indolylpropionic acid can be further converted in the liver or kidney into indolyl acrylic acid (IAcrA) and conjugated with glycine to produce indolylacryloyl glycine (IAcrGly).... Also, indolyl propionic acid has been shown to be a powerful antioxidant, and is currently being investigated as a possible treatment for Alzheimer's disease.40
- ^Poeggeler B, Sambamurti K, Siedlak SL, Perry G, Smith MA, Pappolla MA (April 2010)."A novel endogenous indole protects rodent mitochondria and extends rotifer lifespan".PLOS ONE.5(4): e10206.Bibcode:2010PLoSO...510206P.doi:10.1371/journal.pone.0010206.PMC2858081.PMID20421998.
- ^Karbownik M, Reiter RJ, Garcia JJ, Cabrera J, Burkhardt S, Osuna C, Lewiński A (2001). "Indole-3-propionic acid, a melatonin-related molecule, protects hepatic microsomal membranes from iron-induced oxidative damage: relevance to cancer reduction".Journal of Cellular Biochemistry.81(3): 507–513.doi:10.1002/1097-4644(20010601)81:3<507::AID-JCB1064>3.0.CO;2-M.PMID11255233.S2CID27462000.
- ^Reiter RJ, Tan DX, Osuna C, Gitto E (2000). "Actions of melatonin in the reduction of oxidative stress. A review".Journal of Biomedical Science.7(6): 444–458.doi:10.1007/bf02253360.PMID11060493.