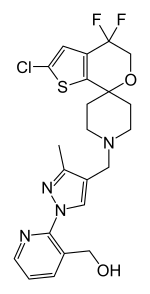
BTRX-246040
Appearance
 | |
| Clinical data | |
|---|---|
| Other names | BTRX-246040 |
| Routes of administration | By mouth |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard(EPA) | |
| Chemical and physical data | |
| Formula | C22H23ClF2N4O2S |
| Molar mass | 480.96g·mol−1 |
| 3D model (JSmol) | |
| |
| |
BTRX-246040,also known asLY-2940094,is apotentandselectivenociceptin receptorantagonistwhich is under development by BlackThorn Therapeutics andEli Lillyfor the treatment ofmajor depressive disorder(MDD).[1][2][3]It has demonstrated proof-of-concept clinical efficacy for depression.[4]As of 2017, it is inphase IIclinical trialsfor the treatment of MDD.[1][2][3]It was also under investigation for the treatment ofalcoholism,and similarly reached phase II clinical studies for this indication, but development was discontinued.[1]
See also[edit]
References[edit]
- ^abc"BTRX 246040".AdisInsight.Springer Nature Switzerland AG.
- ^abDale E, Bang-Andersen B, Sánchez C (May 2015)."Emerging mechanisms and treatments for depression beyond SSRIs and SNRIs".Biochemical Pharmacology.95(2): 81–97.doi:10.1016/j.bcp.2015.03.011.PMID25813654.
- ^abYin X, Guven N, Dietis N (2015)."Opioids in Depression: Not Quite There Yet".UK Journal of Pharmaceutical and Biosciences.3(1): 12–17.doi:10.20510/ukjpb/3/i1/89219.
- ^Post A, Smart TS, Krikke-Workel J, Dawson GR, Harmer CJ, Browning M, et al. (June 2016)."A Selective Nociceptin Receptor Antagonist to Treat Depression: Evidence from Preclinical and Clinical Studies".Neuropsychopharmacology.41(7): 1803–1812.doi:10.1038/npp.2015.348.PMC4869049.PMID26585287.
External links[edit]
- "BTRX-246040 (LY-2940094)".AdisInsight.Springer Nature Switzerland AG.
