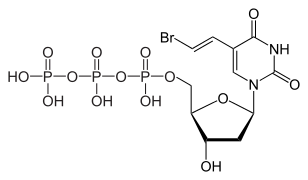
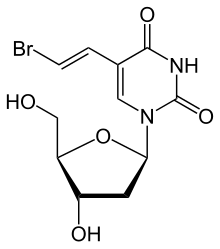
Brivudine
 | |
| Clinical data | |
|---|---|
| Trade names | Zostex, Mevir, Brivir, many others |
| Other names | BVDU |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokineticdata | |
| Bioavailability | 30% |
| Protein binding | >95% |
| Metabolism | Thymidine phosphorylase |
| Metabolites | Bromovinyluracil |
| Eliminationhalf-life | 16 hours |
| Excretion | 65%renal(mainly metabolites), 20%faeces |
| Identifiers | |
| |
| CAS Number | |
| PubChemCID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard(EPA) | |
| Chemical and physical data | |
| Formula | C11H13BrN2O5 |
| Molar mass | 333.138g·mol−1 |
| 3D model (JSmol) | |
| Specific rotation | +9°±1° |
| Density | 1.86 g/cm3 |
| Melting point | 165 to 166 °C (329 to 331 °F) (decomposes) |
| |
| |
Brivudine(trade namesZostex,Mevir,Brivir,among others) is anantiviral drugused in the treatment ofherpes zoster( "shingles" ). Like other antivirals, it acts by inhibitingreplicationof the target virus.
Medical uses
[edit]Brivudine is used for the treatment of herpes zoster in adult patients. It is taken orally once daily, in contrast toaciclovir,valaciclovirand other antivirals.[1]A study has found that it is more effective thanaciclovir,but this has been disputed because of a possibleconflict of intereston part of the study authors.[2]
Contraindications
[edit]The drug is contraindicated in patients undergoingimmunosuppression(for example because of anorgan transplant) or cancer therapy, especially withfluorouracil(5-FU) and chemically related(pro)drugssuch ascapecitabineandtegafur,as well as theantimycoticdrugflucytosine,which is also related to 5-FU. It has not been proven to be safe in children and pregnant or breastfeeding women.[1]
Adverse effects
[edit]The drug is generally well tolerated. The only common side effect isnausea(in 2% of patients). Less common side effects (<1%) include headache, increased or lowered blood cell counts (granulocytopenia,anaemia,lymphocytosis,monocytosis), increased liver enzymes, and allergic reactions.[1]
Interactions
[edit]Brivudine interacts strongly and in rare cases lethally with the anticancer drugfluorouracil(5-FU), its prodrugs and related substances. Eventopicallyapplied 5-FU can be dangerous in combination with brivudine. This is caused by the mainmetabolite,bromovinyluracil (BVU), irreversibly inhibiting the enzymedihydropyrimidine dehydrogenase(DPD) which is necessary for inactivating 5-FU. After a standard brivudine therapy, DPD function can be compromised for up to 18 days. This interaction is shared with the closely related drugsorivudinewhich also has BVU as its main metabolite.[1][3]
There are no other relevant interactions. Brivudine does not significantly influence thecytochrome P450enzymes in the liver.[1]
Pharmacology
[edit]Spectrum of activity
[edit]The drug inhibitsreplicationofvaricella zoster virus(VZV) – which causes herpes zoster – andherpes simplex virustype 1 (HSV-1), but not HSV-2 which typically causes genital herpes.In vitro,inhibitory concentrationsagainst VZV are 200- to 1000-fold lower than those of aciclovir andpenciclovir,theoretically indicating a much higher potency of brivudine. Clinically relevant VZV strains are particularly sensitive.[4]
Mechanism of action
[edit]Brivudine is an analogue of thenucleosidethymidine.The active compound is brivudine 5'-triphosphate, which is formed in subsequentphosphorylationsby viral (but not human)thymidine kinaseand presumably bynucleoside-diphosphate kinase.Brivudine 5'-triphosphate works because it is incorporated into the viral DNA, but then blocks the action ofDNA polymerases,thus inhibiting viral replication.[1][4]
Pharmacokinetics
[edit]Brivudine is well and rapidly absorbed from the gut and undergoesfirst-pass metabolismin the liver, where the enzymethymidine phosphorylase[5]quickly splits off the sugar component, leading to abioavailabilityof 30%. The resulting metabolite is bromovinyluracil (BVU), which does not have antiviral activity. BVU is also the only metabolite that can be detected in the blood plasma.[1][6]
Highest blood plasma concentrations are reached after one hour. Brivudine is almost completely (>95%)bound to plasma proteins.Terminal half-lifeis 16 hours; 65% of the substance are found in the urine and 20% in the faeces, mainly in form of anacetic acidderivative (which is not detectable in the plasma), but also other water-soluble metabolites, which areureaderivatives. Less than 1% is excreted in form of the original compound.[1]
-
Brivudine 5'-triphosphate, theactive metabolite
-
Bromovinyluracil (BVU), the main inactive metabolite
-
Theacetic acidderivative predominantly found in urine
Chemistry
[edit]The molecule has threechiralcarbon atoms in thedeoxyribose(sugar) part all of which have defined orientation; i.e. the drug isstereochemicallypure.[1]The substance is a white powder.
Manufacturing
[edit]Main supplier isBerlin Chemie,now part of Italy'sMenariniGroup. In Central America is provided by Menarini Centro America and Wyeth.[citation needed]
History
[edit]The substance was first synthesized by scientists at theUniversity of Birminghamin the UK in 1976. It was shown to be a potent inhibitor of HSV-1 and VZV byErik De Clercqat theRega Institute for Medical ResearchinBelgiumin 1979. In the 1980s the drug became commercially available inEast Germany,where it was marketed asHelpinby a pharmaceutical company called Berlin-Chemie. Only after the indication was changed to the treatment of herpes zoster in 2001 did it become more widely available in Europe.[7][8]
Brivudine is approved for use in a number of European countries including Austria, Belgium, Germany, Greece, Italy, Portugal, Spain and Switzerland.[9]
Etymology
[edit]The namebrivudinederives from the chemical nomenclaturebromo-vinyl-deoxyuridineor BVDU for short. It is sold under trade names such as Bridic, Brival, Brivex, Brivir, Brivirac, Brivox, Brivuzost, Zerpex, Zonavir, Zostex, and Zovudex.[9]
Research
[edit]ACochrane Systematic Reviewexamined the effectiveness of multiple antiviral drugs in the treatment ofherpes simplex virus epithelial keratitis.Brivudine was found to be significantly more effective thanidoxuridinein increasing the number of successfully healed eyes of participants.[10]
See also
[edit]Related antiviral drugs
- Aciclovir
- Valacyclovir,a prodrug form of aciclovir
- Famciclovir,an analogue of Penciclovir with greater oral availability
- Foscarnet,an intravenous antiviral for aciclovir-resistant VZV
- Penciclovir,a topical preparation
Vaccines and other treatments
- Zostavax,a live virus Herpes zoster (shingles) vaccine
- Varivax,a live virus Varicella Zoster (chickenpox) vaccine
- Shingrix,a recombinant subunit vaccine for shingles
- VZV immune globulin,an antibody-based treatment for immune-suppressed patients with zoster
References
[edit]- ^abcdefghiJasek W, ed. (2007).Austria-Codex(in German) (62nd ed.). Vienna: Österreichischer Apothekerverlag. pp. 5246–8.ISBN978-3-85200-181-4.
- ^"Brivudin (Zostex) besser als Aciclovir (Zovirax a.a.)?".Arznei-telegramm(in German). 5/2007.
- ^"UAW – Aus Fehlern lernen - Potenziell tödlich verlaufende Wechselwirkung zwischen Brivudin (Zostex) und 5-Fluoropyrimidinen"(PDF).Deutsches Ärzteblatt(in German).103(27). 7 July 2006.
- ^abSteinhilber D, Schubert-Zsilavecz M, Roth HJ (2005).Medizinische Chemie(in German). Stuttgart: Deutscher Apotheker Verlag. pp. 581–2.ISBN3-7692-3483-9.
- ^Desgranges C, Razaka G, Rabaud M, Bricaud H, Balzarini J, De Clercq E (December 1983). "Phosphorolysis of (E)-5-(2-bromovinyl)-2'-deoxyuridine (BVDU) and other 5-substituted-2'-deoxyuridines by purified human thymidine phosphorylase and intact blood platelets".Biochemical Pharmacology.32(23): 3583–90.doi:10.1016/0006-2952(83)90307-6.PMID6651877.
- ^Mutschler E, Schäfer-Korting M (2001).Arzneimittelwirkungen(in German) (8 ed.). Stuttgart: Wissenschaftliche Verlagsgesellschaft. p. 847.ISBN3-8047-1763-2.
- ^De Clercq E (December 2004). "Discovery and development of BVDU (brivudin) as a therapeutic for the treatment of herpes zoster".Biochemical Pharmacology.68(12): 2301–15.doi:10.1016/j.bcp.2004.07.039.PMID15548377.
- ^Tringali C, ed. (2012).Bioactive Compounds from Natural Sources(2nd ed.). CRC Press. p.170.
- ^ab"Brivudine".drugs.com.
- ^Wilhelmus KR (January 2015)."Antiviral treatment and other therapeutic interventions for herpes simplex virus epithelial keratitis".The Cochrane Database of Systematic Reviews.1(1): CD002898.doi:10.1002/14651858.CD002898.pub5.PMC4443501.PMID25879115.