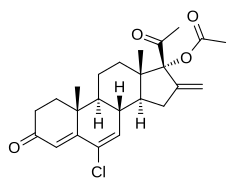
Chlormethenmadinone acetate
Appearance
 | |
| Clinical data | |
|---|---|
| Trade names | Biogest, Sterolibrin, Antigest B, Agelin |
| Other names | SCH-12600; 6-Chloromethylenedehydroacetoxyprogesterone; 17α-Acetoxy-6-chloro-16-methylene-6-dehydroprogesterone; 16-Methylenechlormadinone acetate; 17α-Acetoxy-6-chloro-16-methylenepregna-4,6-diene-3,20-dione |
| Drug class | Progestogen;Progestin;Progestogen ester |
| Identifiers | |
| |
| CAS Number | |
| PubChemCID | |
| ChemSpider | |
| ChEMBL | |
| CompTox Dashboard(EPA) | |
| Chemical and physical data | |
| Formula | C24H29ClO4 |
| Molar mass | 416.94g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Chlormethenmadinone acetate(CMMA), also known aschlorsuperlutin,is aprogestinmedication which was developed inCzechoslovakiain the 1960s.[1]It has been used in combination withmestranolinbirth control pillsunder the brand namesBiogest,Sterolibrin,andAntigest B,[2][3]and inveterinary medicineunder the brand nameAgelin.[4]Analoguesof CMMA includebromethenmadinone acetate(bromsuperlutin), which was assessed but was never marketed,[3][5]andmelengestrol acetate(methylsuperlutin), which is used in veterinary medicine.[6]
See also[edit]
- List of progestogen esters § Esters of 17α-hydroxyprogesterone derivatives
- 16-Methylene-17α-hydroxyprogesterone acetate
References[edit]
- ^Sterba R (1968)."New biological application of contraceptive steroids".Endocrinologia Experimentalis.2(2): 101–110. Archived fromthe originalon 16 September 2018.
- ^Melich H (July 1972)."[Biogest]".Casopis Lekaru Ceskych(in Czech).111(30): 694–695.PMID5079918.Archived fromthe originalon 2018-09-16.Retrieved2018-09-16.
- ^abStĕrba R (March 1970)."[Towards a more physiological hormonal contraception]".Zentralblatt Fur Gynakologie(in German).92(10): 303–312.PMID4096927.Archived fromthe originalon 2018-09-16.Retrieved2018-09-16.
- ^Bekeová E, Krajnicáková M, Hendrichovský V, Maracek I (November 1995)."[Thyroid and ovarian hormones in ewes treated with gestagens and PMSG in the spring season]".Veterinarni Medicina(in Slovak).40(11): 345–352.PMID8659087.Archived fromthe originalon 2018-09-16.Retrieved2018-09-16.
- ^Štěrba, R. (1971)."On the Way to a More Physiological Hormonal Contraception".Current Problems in Fertility.pp. 154–158.doi:10.1007/978-1-4615-8651-7_28.ISBN978-1-4615-8653-1.Archived fromthe originalon 2018-09-16.Retrieved2018-09-16.
- ^von Kunz W (8 March 2013)."Über neue Arzneimittel".In Denkewalter RG, Tishler M, Ehrhart G, Biel JH, Lum BK, Büchi J, Winter CA, Münzel K, Kunz W, Ariëns EJ, Labhardt F (eds.).Fortschritte der Arzneimittelforschung / Progress in Drug Research / Progrès des recherches pharmaceutiques.Birkhäuser. pp. 407–.ISBN978-3-0348-7059-7.
