Curie temperature
Inphysicsandmaterials science,theCurie temperature(TC), orCurie point,is the temperature above which certain materials lose theirpermanent magneticproperties, which can (in most cases) be replaced byinduced magnetism.The Curie temperature is named afterPierre Curie,who showed that magnetism was lost at a critical temperature.[1]
The force of magnetism is determined by themagnetic moment,a dipole moment within an atom that originates from theangular momentumandspinof electrons. Materials have different structures of intrinsic magnetic moments that depend on temperature; the Curie temperature is the critical point at which a material's intrinsic magnetic moments change direction.
Permanent magnetism is caused by the alignment of magnetic moments, and induced magnetism is created when disordered magnetic moments are forced to align in an applied magnetic field. For example, the ordered magnetic moments (ferromagnetic,Figure 1) change and become disordered (paramagnetic,Figure 2) at the Curie temperature. Higher temperatures make magnets weaker, as spontaneous magnetism only occurs below the Curie temperature.Magnetic susceptibilityabove the Curie temperature can be calculated from theCurie–Weiss law,which is derived fromCurie's law.
In analogy to ferromagnetic and paramagnetic materials, the Curie temperature can also be used to describe the phase transition betweenferroelectricityandparaelectricity.In this context, theorder parameteris theelectricpolarizationthat goes from a finite value to zero when the temperature is increased above the Curie temperature.
| Material | Curie temperature (K) |
°C | °F |
|---|---|---|---|
| Iron(Fe) | 1043-1,664 | 770 | 1418 |
| Cobalt(Co) | 1400 | 1130 | 2060 |
| Nickel(Ni) | 627 | 354 | 669 |
| Gadolinium(Gd) | 292 | 19 | 66 |
| Dysprosium(Dy) | 88 | −185.2 | −301.3 |
| Manganese bismuthide(MnBi) | 630 | 357 | 674 |
| Manganese antimonide (MnSb) | 587 | 314 | 597 |
| Chromium(IV) oxide(CrO2) | 386 | 113 | 235 |
| Manganese arsenide (MnAs) | 318 | 45 | 113 |
| Europium oxide (EuO) | 69 | −204.2 | −335.5 |
| Iron(III) oxide (Fe2O3) | 948 | 675 | 1247 |
| Iron(II,III) oxide(FeOFe2O3) | 858 | 585 | 1085 |
| NiO–Fe2O3 | 858 | 585 | 1085 |
| CuO–Fe2O3 | 728 | 455 | 851 |
| MgO–Fe2O3 | 713 | 440 | 824 |
| MnO–Fe2O3 | 573 | 300 | 572 |
| Yttrium iron garnet(Y3Fe5O12) | 560 | 287 | 548 |
| Neodymium magnets | 583–673 | 310–400 | 590–752 |
| Alnico | 973–1133 | 700–860 | 1292–1580 |
| Samarium–cobalt magnets | 993–1073 | 720–800 | 1328–1472 |
| Strontium ferrite | 723 | 450 | 842 |
History
[edit]That heating destroys magnetism was already described inDe Magnete(1600):
Iron filings, after being heated for a long time, are attracted by a loadstone, yet not so strongly or from so great a distance as when not heated. A loadstone loses some of its virtue by too great a heat; for its humour is set free, whence its peculiar nature is marred. (Book 2, Chapter 23).
Magnetic moments
[edit]At the atomic level, there are two contributors to themagnetic moment,theelectron magnetic momentand thenuclear magnetic moment.Of these two terms, the electron magnetic moment dominates, and the nuclear magnetic moment is insignificant. At higher temperatures, electrons have higher thermal energy. This has a randomizing effect on aligned magnetic domains, leading to the disruption of order, and the phenomena of the Curie point.[5][6]
Ferromagnetic,paramagnetic,ferrimagnetic,andantiferromagneticmaterials have different intrinsic magnetic moment structures. At a material's specific Curie temperature (TC), these properties change. The transition from antiferromagnetic to paramagnetic (or vice versa) occurs at theNéel temperature(TN), which is analogous to Curie temperature.
| BelowTC | AboveTC |
|---|---|
| Ferromagnetic | ↔ Paramagnetic |
| Ferrimagnetic | ↔ Paramagnetic |
| BelowTN | AboveTN |
| Antiferromagnetic | ↔ Paramagnetic |
- Orientations of magnetic moments in materials
-
Ferromagnetism:The magnetic moments in a ferromagnetic material are ordered and of the same magnitude in the absence of an applied magnetic field.
-
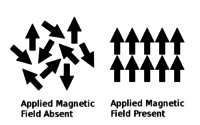
Paramagnetism:The magnetic moments in a paramagnetic material are disordered in the absence of an applied magnetic field and ordered in the presence of an applied magnetic field.
-
Ferrimagnetism:The magnetic moments in a ferrimagnetic material have different magnitudes (due to the crystal containing two different types of magnetic ions[clarification needed]) which are aligned oppositely in the absence of an applied magnetic field.
-
Antiferromagnetism:The magnetic moments in an antiferromagnetic material have the same magnitudes but are aligned oppositely in the absence of an applied magnetic field.
Materials with magnetic moments that change properties at the Curie temperature
[edit]Ferromagnetic, paramagnetic, ferrimagnetic, and antiferromagnetic structures are made up of intrinsic magnetic moments. If all the electrons within the structure are paired, these moments cancel out due to their opposite spins and angular momenta. Thus, even with an applied magnetic field, these materials have different properties and no Curie temperature.[7][8]
Paramagnetic
[edit]A material is paramagnetic only above its Curie temperature. Paramagnetic materials are non-magnetic when amagnetic fieldis absent and magnetic when a magnetic field is applied. When a magnetic field is absent, the material has disordered magnetic moments; that is, the magnetic moments are asymmetrical and not aligned. When a magnetic field is present, the magnetic moments are temporarily realigned parallel to the applied field;[9][10]the magnetic moments are symmetrical and aligned.[11]The magnetic moments being aligned in the same direction are what causes an induced magnetic field.[11][12]
For paramagnetism, this response to an applied magnetic field is positive and is known asmagnetic susceptibility.[7]The magnetic susceptibility only applies above the Curie temperature for disordered states.[13]
Sources of paramagnetism (materials which have Curie temperatures) include:[14]
- All atoms that have unpaired electrons;
- Atoms that have inner shells that are incomplete in electrons;
- Free radicals;
- Metals.
Above the Curie temperature, the atoms are excited, and the spin orientations become randomized[8]but can be realigned by an applied field, i.e., the material becomes paramagnetic. Below the Curie temperature, the intrinsic structure has undergone aphase transition,[15]the atoms are ordered, and the material is ferromagnetic.[11]The paramagnetic materials' induced magnetic fields are very weak compared with ferromagnetic materials' magnetic fields.[15]
Ferromagnetic
[edit]Materials are only ferromagnetic below their corresponding Curie temperatures. Ferromagnetic materials are magnetic in the absence of an applied magnetic field.
When a magnetic field is absent the material hasspontaneous magnetizationwhich is a result of the ordered magnetic moments; that is, for ferromagnetism, the atoms are symmetrical and aligned in the same direction creating a permanent magnetic field.
The magnetic interactions are held together byexchange interactions;otherwise thermal disorder would overcome the weak interactions of magnetic moments. The exchange interaction has a zero probability of parallel electrons occupying the same point in time, implying a preferred parallel alignment in the material.[16]The Boltzmann factor contributes heavily as it prefers interacting particles to be aligned in the same direction.[17]This causesferromagnetsto have strong magnetic fields and high Curie temperatures of around 1,000 K (730 °C).[18]
Below the Curie temperature, the atoms are aligned and parallel, causing spontaneous magnetism; the material is ferromagnetic. Above the Curie temperature the material is paramagnetic, as the atoms lose their ordered magnetic moments when the material undergoes a phase transition.[15]
Ferrimagnetic
[edit]Materials are only ferrimagnetic below their corresponding Curie temperature. Ferrimagnetic materials are magnetic in the absence of an applied magnetic field and are made up of two differentions.[19]
When a magnetic field is absent the material has a spontaneous magnetism which is the result of ordered magnetic moments; that is, for ferrimagnetism one ion's[clarification needed]magnetic moments are aligned facing in one direction with certain magnitude and the other ion's magnetic moments are aligned facing in the opposite direction with a different magnitude. As the magnetic moments are of different magnitudes in opposite directions there is still a spontaneous magnetism and a magnetic field is present.[19]
Similar to ferromagnetic materials the magnetic interactions are held together by exchange interactions. The orientations of moments however are anti-parallel which results in a net momentum by subtracting their momentum from one another.[19]
Below the Curie temperature the atoms of each ion are aligned anti-parallel with different momentums causing a spontaneous magnetism; the material is ferrimagnetic. Above the Curie temperature the material is paramagnetic as the atoms lose their ordered magnetic moments as the material undergoes a phase transition.[19]
Antiferromagnetic and the Néel temperature
[edit]Materials are only antiferromagnetic below their correspondingNéel temperatureormagnetic ordering temperature,TN.This is similar to the Curie temperature as above the Néel Temperature the material undergoes aphase transitionand becomes paramagnetic. That is, the thermal energy becomes large enough to destroy the microscopic magnetic ordering within the material.[20]It is named afterLouis Néel(1904–2000), who received the 1970Nobel Prize in Physicsfor his work in the area.
The material has equal magnetic moments aligned in opposite directions resulting in a zero magnetic moment and a net magnetism of zero at all temperatures below the Néel temperature. Antiferromagnetic materials are weakly magnetic in the absence or presence of an applied magnetic field.
Similar to ferromagnetic materials the magnetic interactions are held together by exchange interactions preventing thermal disorder from overcoming the weak interactions of magnetic moments.[16][21]When disorder occurs it is at the Néel temperature.[21]
Listed below are the Néel temperatures of several materials:[22]
| Substance | Néel temperature (K) |
|---|---|
| MnO | 116 |
| MnS | 160 |
| MnTe | 307 |
| MnF2 | 67 |
| FeF2 | 79 |
| FeCl2 | 24 |
| FeI2 | 9 |
| FeO | 198 |
| FeOCl | 80 |
| CrCl2 | 25 |
| CrI2 | 12 |
| CoO | 291 |
| NiCl2 | 50 |
| NiI2 | 75 |
| NiO | 525 |
| KFeO2 | 983[23] |
| Cr | 308 |
| Cr2O3 | 307 |
| Nd5Ge3 | 50 |
Curie–Weiss law
[edit]The Curie–Weiss law is an adapted version ofCurie's law.
The Curie–Weiss law is a simple model derived from amean-fieldapproximation, this means it works well for the materials temperature,T,much greater than their corresponding Curie temperature,TC,i.e.T≫TC;however fails to describe themagnetic susceptibility,χ,in the immediate vicinity of the Curie point because of local fluctuations between atoms.[24]
Neither Curie's law nor the Curie–Weiss law holds forT<TC.
Curie's law for a paramagnetic material:[25]
| Definition | |
|---|---|
| χ | the magnetic susceptibility; the influence of an appliedmagnetic fieldon a material |
| M | themagnetic momentsper unit volume |
| H | the macroscopic magnetic field |
| B | the magnetic field |
| C | the material-specificCurie constant |
| theAvogadro constant | |
| µ0 | thepermeability of free space.Note: inCGSunits is taken to equal one.[27] |
| g | theLandég-factor |
| J(J+ 1) | the eigenvalue for eigenstate J2for the stationary states within the incomplete atoms shells (electrons unpaired) |
| µB | theBohr magneton |
| kB | theBoltzmann constant |
| total magnetism | isNnumber of magnetic moments per unit volume |
The Curie–Weiss law is then derived from Curie's law to be:
where:
λis the Weiss molecular field constant.[26][28]
For full derivation seeCurie–Weiss law.
Physics
[edit]Approaching Curie temperature from above
[edit]As the Curie–Weiss law is an approximation, a more accurate model is needed when the temperature,T,approaches the material's Curie temperature,TC.
Magnetic susceptibility occurs above the Curie temperature.
An accurate model of critical behaviour for magnetic susceptibility withcritical exponentγ:
The critical exponent differs between materials and for themean-fieldmodel is taken asγ= 1.[29]
As temperature is inversely proportional to magnetic susceptibility, whenTapproachesTCthe denominator tends to zero and the magnetic susceptibility approachesinfinityallowing magnetism to occur. This is a spontaneous magnetism which is a property of ferromagnetic and ferrimagnetic materials.[30][31]
Approaching Curie temperature from below
[edit]Magnetism depends on temperature and spontaneous magnetism occurs below the Curie temperature. An accurate model of critical behaviour for spontaneous magnetism with critical exponentβ:
The critical exponent differs between materials and for the mean-field model as taken asβ=1/2whereT≪TC.[29]
The spontaneous magnetism approaches zero as the temperature increases towards the materials Curie temperature.
Approaching absolute zero (0 kelvin)
[edit]The spontaneous magnetism, occurring in ferromagnetic, ferrimagnetic, and antiferromagnetic materials, approaches zero as the temperature increases towards the material's Curie temperature. Spontaneous magnetism is at its maximum as the temperature approaches0 K.[32]That is, the magnetic moments are completely aligned and at their strongest magnitude of magnetism due to lack of thermal disturbance.
In paramagnetic materials thermal energy is sufficient to overcome the ordered alignments. As the temperature approaches 0 K, theentropydecreases to zero, that is, the disorder decreases and the material becomes ordered. This occurs without the presence of an applied magnetic field and obeys thethird law of thermodynamics.[16]
Both Curie's law and the Curie–Weiss law fail as the temperature approaches 0 K. This is because they depend on the magnetic susceptibility, which only applies when the state is disordered.[33]
Gadolinium sulfatecontinues to satisfy Curie's law at 1 K. Between 0 and 1 K the law fails to hold and a sudden change in the intrinsic structure occurs at the Curie temperature.[34]
Ising model of phase transitions
[edit]TheIsing modelis mathematically based and can analyse the critical points ofphase transitionsin ferromagnetic order due to spins of electrons having magnitudes of ±1/2.The spins interact with their neighbouringdipoleelectrons in the structure and here the Ising model can predict their behaviour with each other.[35][36]
This model is important for solving and understanding the concepts of phase transitions and hence solving the Curie temperature. As a result, many different dependencies that affect the Curie temperature can be analysed.
For example, the surface and bulk properties depend on the alignment and magnitude of spins and the Ising model can determine the effects of magnetism in this system.
One should note, in 1D the Curie (critical) temperature for a magnetic order phase transition is found to be at zero temperature, i.e. the magnetic order takes over only at T = 0. In 2D, the critical temperature, e.g. a finite magnetization, can be calculated by solving the inequality:
Weiss domains and surface and bulk Curie temperatures
[edit]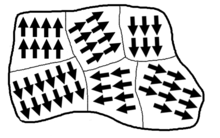
Materials structures consist of intrinsic magnetic moments which are separated into domains calledWeiss domains.[37]This can result in ferromagnetic materials having no spontaneous magnetism as domains could potentially balance each other out.[37]The position of particles can therefore have different orientations around the surface than the main part (bulk) of the material. This property directly affects the Curie temperature as there can be a bulk Curie temperatureTBand a different surface Curie temperatureTSfor a material.[38]
This allows for the surface Curie temperature to be ferromagnetic above the bulk Curie temperature when the main state is disordered, i.e. Ordered and disordered states occur simultaneously.[35]
The surface and bulk properties can be predicted by the Ising model and electron capture spectroscopy can be used to detect the electron spins and hence themagnetic momentson the surface of the material. An average total magnetism is taken from the bulk and surface temperatures to calculate the Curie temperature from the material, noting the bulk contributes more.[35][39]
Theangular momentumof an electron is either +ħ/2or −ħ/2due to it having a spin of1/2,which gives a specific size of magnetic moment to the electron; theBohr magneton.[40]Electrons orbiting around the nucleus in a current loop create a magnetic field which depends on the Bohr magneton andmagnetic quantum number.[40]Therefore, the magnetic moments are related between angular and orbital momentum and affect each other. Angular momentum contributes twice as much to magnetic moments than orbital.[41]
Forterbiumwhich is arare-earth metaland has a high orbital angular momentum the magnetic moment is strong enough to affect the order above its bulk temperatures. It is said to have a highanisotropyon the surface, that is it is highly directed in one orientation. It remains ferromagnetic on its surface above its Curie temperature (219K) while its bulk becomes antiferromagnetic and then at higher temperatures its surface remains antiferromagnetic above its bulk Néel Temperature (230K) before becoming completely disordered and paramagnetic with increasing temperature. The anisotropy in the bulk is different from its surface anisotropy just above these phase changes as the magnetic moments will be ordered differently or ordered in paramagnetic materials.[38][42]
Changing a material's Curie temperature
[edit]Composite materials
[edit]Composite materials,that is, materials composed from other materials with different properties, can change the Curie temperature. For example, a composite which hassilverin it can create spaces for oxygen molecules in bonding which decreases the Curie temperature[43]as the crystal lattice will not be as compact.
The alignment of magnetic moments in the composite material affects the Curie temperature. If the material's moments are parallel with each other, the Curie temperature will increase and if perpendicular the Curie temperature will decrease[43]as either more or less thermal energy will be needed to destroy the alignments.
Preparing composite materials through different temperatures can result in different final compositions which will have different Curie temperatures.[44]Dopinga material can also affect its Curie temperature.[44]
The density of nanocomposite materials changes the Curie temperature.Nanocompositesare compact structures on a nano-scale. The structure is built up of high and low bulk Curie temperatures, however will only have one mean-field Curie temperature. A higher density of lower bulk temperatures results in a lower mean-field Curie temperature, and a higher density of higher bulk temperature significantly increases the mean-field Curie temperature. In more than one dimension the Curie temperature begins to increase as the magnetic moments will need more thermal energy to overcome the ordered structure.[39]
Particle size
[edit]The size of particles in a material's crystal lattice changes the Curie temperature. Due to the small size of particles (nanoparticles) the fluctuations of electron spins become more prominent, which results in the Curie temperature drastically decreasing when the size of particles decreases, as the fluctuations cause disorder. The size of a particle also affects theanisotropycausing alignment to become less stable and thus lead to disorder in magnetic moments.[35][45]
The extreme of this issuperparamagnetismwhich only occurs in small ferromagnetic particles. In this phenomenon, fluctuations are very influential causing magnetic moments to change direction randomly and thus create disorder.
The Curie temperature of nanoparticles is also affected by thecrystal latticestructure:body-centred cubic(bcc),face-centred cubic(fcc), and ahexagonalstructure (hcp) all have different Curie temperatures due to magnetic moments reacting to their neighbouring electron spins. fcc and hcp have tighter structures and as a results have higher Curie temperatures than bcc as the magnetic moments have stronger effects when closer together.[35]This is known as thecoordination numberwhich is the number of nearest neighbouring particles in a structure. This indicates a lower coordination number at the surface of a material than the bulk which leads to the surface becoming less significant when the temperature is approaching the Curie temperature. In smaller systems the coordination number for the surface is more significant and the magnetic moments have a stronger effect on the system.[35]
Although fluctuations in particles can be minuscule, they are heavily dependent on the structure of crystal lattices as they react with their nearest neighbouring particles. Fluctuations are also affected by the exchange interaction[45]as parallel facing magnetic moments are favoured and therefore have less disturbance and disorder, therefore a tighter structure influences a stronger magnetism and therefore a higher Curie temperature.
Pressure
[edit]Pressure changes a material's Curie temperature. Increasingpressureon thecrystal latticedecreases the volume of the system. Pressure directly affects thekinetic energyin particles as movement increases causing the vibrations to disrupt the order of magnetic moments. This is similar to temperature as it also increases the kinetic energy of particles and destroys the order of magnetic moments and magnetism.[46]
Pressure also affects thedensity of states(DOS).[46]Here the DOS decreases causing the number of electrons available to the system to decrease. This leads to the number of magnetic moments decreasing as they depend on electron spins. It would be expected because of this that the Curie temperature would decrease; however, it increases. This is the result of theexchange interaction.The exchange interaction favours the aligned parallel magnetic moments due to electrons being unable to occupy the same space in time[16]and as this is increased due to the volume decreasing the Curie temperature increases with pressure. The Curie temperature is made up of a combination of dependencies on kinetic energy and the DOS.[46]
The concentration of particles also affects the Curie temperature when pressure is being applied and can result in a decrease in Curie temperature when the concentration is above a certain percent.[46]
Orbital ordering
[edit]Orbital orderingchanges the Curie temperature of a material. Orbital ordering can be controlled through appliedstrains.[47]This is a function that determines the wave of a single electron or paired electrons inside the material. Having control over theprobabilityof where the electron will be allows the Curie temperature to be altered. For example, thedelocalisedelectrons can be moved onto the sameplaneby applied strains within the crystal lattice.[47]
The Curie temperature is seen to increase greatly due to electrons being packed together in the same plane, they are forced to align due to theexchange interactionand thus increases the strength of the magnetic moments which prevents thermal disorder at lower temperatures.
Curie temperature in ferroelectric materials
[edit]In analogy to ferromagnetic and paramagnetic materials, the term Curie temperature (TC) is also applied to the temperature at which aferroelectricmaterial transitions to beingparaelectric.Hence,TCis the temperature where ferroelectric materials lose their spontaneous polarisation as a first or second order phase change occurs. In case of a second order transition, the Curie Weiss temperatureT0which defines the maximum of the dielectric constant is equal to the Curie temperature. However, the Curie temperature can be 10 K higher thanT0in case of a first order transition.[48]
| BelowTC | AboveTC[49] |
|---|---|
| Ferroelectric | ↔ Dielectric (paraelectric) |
| Antiferroelectric | ↔ Dielectric (paraelectric) |
| Ferrielectric | ↔ Dielectric (paraelectric) |
| Helielectric | ↔ Dielectric (paraelectric) |
Ferroelectric and dielectric
[edit]Materials are only ferroelectric below their corresponding transition temperatureT0.[50]Ferroelectric materials are allpyroelectricand therefore have a spontaneous electric polarisation as the structures are unsymmetrical.
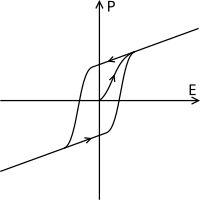
Ferroelectric materials' polarization is subject tohysteresis(Figure 4); that is they are dependent on their past state as well as their current state. As an electric field is applied the dipoles are forced to align and polarisation is created, when the electric field is removed polarisation remains. The hysteresis loop depends on temperature and as a result as the temperature is increased and reachesT0the two curves become one curve as shown in the dielectric polarisation (Figure 5).[51]
Relative permittivity
[edit]A modified version of the Curie–Weiss law applies to the dielectric constant, also known as therelative permittivity:[48][52]
Applications
[edit]A heat-induced ferromagnetic-paramagnetic transition is used inmagneto-opticalstorage media for erasing and writing of new data. Famous examples include theSony Minidiscformat as well as the now-obsoleteCD-MOformat. Curie point electro-magnets have been proposed and tested for actuation mechanisms in passive safety systems offast breeder reactors,wherecontrol rodsare dropped into the reactor core if the actuation mechanism heats up beyond the material's Curie point.[53]Other uses include temperature control insoldering irons[54]and stabilizing the magnetic field oftachometergenerators against temperature variation.[55]
See also
[edit]- Ferroelectricity– Characteristic of certain crystalline materials
- Curie's law– Relation of magnetization to applied magnetic field and temperature
- Hopkinson effect– Feature of ferromagnetic or ferrimagnetic materials
Notes
[edit]- ^Pierre Curie – Biography
- ^Buschow 2001,p5021, table 1
- ^Jullien & Guinier 1989,p. 155
- ^Kittel 1986
- ^Hall & Hook 1994,p. 200
- ^Jullien & Guinier 1989,pp. 136–38
- ^abIbach & Lüth 2009
- ^abLevy 1968,pp. 236–39
- ^Dekker 1958,pp. 217–20
- ^Levy 1968
- ^abcFan 1987,pp. 164–65
- ^Dekker 1958,pp. 454–55
- ^Mendelssohn 1977,p. 162
- ^Levy 1968,pp. 198–202
- ^abcCusack 1958,p. 269
- ^abcdHall & Hook 1994,pp. 220–21
- ^Palmer 2007
- ^Hall & Hook 1994,p. 220
- ^abcdJullien & Guinier 1989,pp. 158–59
- ^Spaldin, Nicola A.(2006).Magnetic materials: fundamentals and device applications(Repr. ed.). Cambridge: Cambridge Univ. Press. pp. 89–106.ISBN9780521016582.
- ^abJullien & Guinier 1989,pp. 156–57
- ^Kittel, Charles (2005).Introduction to Solid State Physics(8th ed.). New York: John Wiley & Sons.ISBN978-0-471-41526-8.
- ^Ichida, Toshio (1973)."Mössbauer Study of the Thermal Decomposition Products of K2FeO4".Bulletin of the Chemical Society of Japan.46(1): 79–82.doi:10.1246/bcsj.46.79.
- ^Jullien & Guinier 1989,pp. 153
- ^Hall & Hook 1994,pp. 205–06
- ^abLevy 1968,pp. 201–02
- ^Kittel 1996,p. 444
- ^Myers 1997,pp. 334–45
- ^abHall & Hook 1994,pp. 227–28
- ^Kittel 1986,pp. 424–26
- ^Spaldin 2010,pp. 52–54
- ^Hall & Hook 1994,p. 225
- ^Mendelssohn 1977,pp. 180–81
- ^Mendelssohn 1977,p. 167
- ^abcdefBertoldi, Bringa & Miranda 2012
- ^Brout 1965,pp. 6–7
- ^abJullien & Guinier 1989,p. 161
- ^abRau, Jin & Robert 1988
- ^abSkomski & Sellmyer 2000
- ^abJullien & Guinier 1989,p. 138
- ^Hall & Hook 1994
- ^Jackson, M. (2000)."Magnetism of Rare Earth"(PDF).The IRM Quarterly.10(3): 1. Archived fromthe original(PDF)on 12 July 2017.Retrieved21 January2020.
- ^abHwang et al. 1998
- ^abPaulsen et al. 2003
- ^abLópez Domínguez et al. 2013
- ^abcdBose et al. 2011
- ^abSadoc et al. 2010
- ^abWebster 1999
- ^Kovetz 1990,p. 116
- ^Myers 1997,pp. 404–05
- ^Pascoe 1973,pp. 190–91
- ^Webster 1999,pp. 6.55–6.56
- ^Takamatsu (2007)."Demonstration of Control Rod Holding Stability of the Self Actuated Shutdown System in Joyo for Enhancement of Fast Reactor Inherent Safety".Journal of Nuclear Science and Technology.44(3): 511–517.doi:10.1080/18811248.2007.9711316.
- ^TMT-9000S
- ^Pallàs-Areny & Webster 2001,pp. 262–63
References
[edit]- Buschow, K. H. J. (2001).Encyclopedia of Materials: Science and Technology.Elsevier.ISBN0-08-043152-6.
- Kittel, Charles(1986).Introduction to Solid State Physics(6th ed.).John Wiley & Sons.ISBN0-471-87474-4.
- Pallàs-Areny, Ramon; Webster, John G. (2001).Sensors and Signal Conditioning(2nd ed.).John Wiley & Sons.ISBN978-0-471-33232-9.
- Spaldin, Nicola A.(2010).Magnetic Materials: Fundamentals and Applications(2nd ed.). Cambridge: Cambridge University Press.ISBN9780521886697.
- Ibach, Harald; Lüth, Hans (2009).Solid-State Physics: An Introduction to Principles of Materials Science(4th ed.). Berlin: Springer.ISBN9783540938033.
- Levy, Robert A. (1968).Principles of Solid State Physics.Academic Press.ISBN978-0124457508.
- Fan, H. Y. (1987).Elements of Solid State Physics.Wiley-Interscience.ISBN9780471859871.
- Dekker, Adrianus J. (1958).Solid State Physics.Macmillan.ISBN9780333106235.
- Cusack, N. (1958).The Electrical and Magnetic Properties of Solids.Longmans, Green.
- Hall, J. R.; Hook, H. E. (1994).Solid State Physics(2nd ed.). Chichester: Wiley.ISBN0471928054.
- Jullien, André; Guinier, Rémi (1989).The Solid State from Superconductors to Superalloys.Oxford: Oxford Univ. Press.ISBN0198555547.
- Mendelssohn, K. (1977).The Quest for Absolute Zero: The Meaning of Low Temperature Physics.with S.I. units. (2nd ed.). London: Taylor and Francis.ISBN0850661196.
- Myers, H. P. (1997).Introductory Solid State Physics(2nd ed.). London: Taylor & Francis.ISBN0748406603.
- Kittel, Charles (1996).Introduction to Solid State Physics(7th ed.). New York [u.a.]: Wiley.ISBN0471111813.
- Palmer, John (2007).Planar Ising correlations(Online ed.). Boston: Birkhäuser.ISBN9780817646202.
- Bertoldi, Dalía S.; Bringa, Eduardo M.; Miranda, E. N. (May 2012)."Analytical solution of the mean field Ising model for finite systems".Journal of Physics: Condensed Matter.24(22): 226004.Bibcode:2012JPCM...24v6004B.doi:10.1088/0953-8984/24/22/226004.hdl:11336/16945.PMID22555147.S2CID34323416.Retrieved12 February2013.
- Brout, Robert (1965).Phase Transitions.New York, Amsterdam: W. A. Benjamin, Inc.
- Rau, C.; Jin, C.; Robert, M. (1988). "Ferromagnetic order at Tb surfaces above the bulk Curie temperature".Journal of Applied Physics.63(8): 3667.Bibcode:1988JAP....63.3667R.doi:10.1063/1.340679.
- Skomski, R.; Sellmyer, D. J. (2000)."Curie temperature of multiphase nanostructures".Journal of Applied Physics.87(9): 4756.Bibcode:2000JAP....87.4756S.doi:10.1063/1.373149.S2CID288790.
- López Domínguez, Victor; Hernàndez, Joan Manel; Tejada, Javier; Ziolo, Ronald F. (14 November 2012). "Colossal Reduction in Curie Temperature Due to Finite-Size Effects inCoFe
2O
4Nanoparticles ".Chemistry of Materials.25(1): 6–11.doi:10.1021/cm301927z. - Bose, S. K.; Kudrnovský, J.; Drchal, V.; Turek, I. (18 November 2011). "Pressure dependence of Curie temperature and resistivity in complex Heusler alloys".Physical Review B.84(17): 174422.arXiv:1010.3025.Bibcode:2011PhRvB..84q4422B.doi:10.1103/PhysRevB.84.174422.S2CID118595011.
- Webster, John G., ed. (1999).The Measurement, Instrumentation, and Sensors Handbook(Online ed.). Boca Raton, FL: CRC Press published in cooperation with IEEE Press.ISBN0849383471.
- Whatmore, R. W. (1991).Electronic Materials: From Silicon to Organics(2nd ed.). New York, NY: Springer.ISBN978-1-4613-6703-1.
- Kovetz, Attay (1990).The Principles of Electromagnetic Theory(1st ed.). Cambridge, UK: Cambridge University Press.ISBN0-521-39997-1.
- Hummel, Rolf E. (2001).Electronic Properties of Materials(3rd ed.). New York [u.a.]: Springer.ISBN0-387-95144-X.
- Pascoe, K. J. (1973).Properties of Materials for Electrical Engineers.New York, N.Y.: J. Wiley and Sons.ISBN0471669113.
- Paulsen, J. A.; Lo, C. C. H.; Snyder, J. E.; Ring, A. P.; Jones, L. L.; Jiles, D. C. (23 September 2003). "Study of the Curie temperature of cobalt ferrite based composites for stress sensor applications".IEEE Transactions on Magnetics.39(5): 3316–18.Bibcode:2003ITM....39.3316P.doi:10.1109/TMAG.2003.816761.ISSN0018-9464.S2CID45734431.
- Hwang, Hae Jin; Nagai, Toru; Ohji, Tatsuki; Sando, Mutsuo; Toriyama, Motohiro; Niihara, Koichi (March 1998). "Curie temperature Anomaly in Lead Zirconate Titanate/Silver Composites".Journal of the American Ceramic Society.81(3): 709–12.doi:10.1111/j.1151-2916.1998.tb02394.x.
- Sadoc, Aymeric; Mercey, Bernard; Simon, Charles; Grebille, Dominique; Prellier, Wilfrid; Lepetit, Marie-Bernadette (2010). "Large Increase of the Curie temperature by Orbital Ordering Control".Physical Review Letters.104(4): 046804.arXiv:0910.3393.Bibcode:2010PhRvL.104d6804S.doi:10.1103/PhysRevLett.104.046804.PMID20366729.S2CID35041713.
- Kochmański, Martin; Paszkiewicz, Tadeusz; Wolski, Sławomir (2013). "Curie–Weiss magnet: a simple model of phase transition".European Journal of Physics.34(6): 1555–73.arXiv:1301.2141.Bibcode:2013EJPh...34.1555K.doi:10.1088/0143-0807/34/6/1555.S2CID118331770.
- "Pierre Curie – Biography".Nobelprize.org.Nobel Media AB. 2014.Retrieved14 March2013.
- "TMT-9000S Soldering and Rework Station".thermaltronics.com.Retrieved13 January2016.




![Ferrimagnetism: The magnetic moments in a ferrimagnetic material have different magnitudes (due to the crystal containing two different types of magnetic ions[clarification needed]) which are aligned oppositely in the absence of an applied magnetic field.](https://upload.wikimedia.org/wikipedia/commons/thumb/e/e9/Diagram_of_Ferrimagnetic_Magnetic_Moments.png/288px-Diagram_of_Ferrimagnetic_Magnetic_Moments.png)