Endocrine disruptor
This articleneeds morereliable medical referencesforverificationor relies too heavily onprimary sources.(August 2019) |  |

Endocrine disruptors,sometimes also referred to ashormonally active agents,[1]endocrine disrupting chemicals,[2]orendocrine disrupting compounds[3]are chemicals that can interfere withendocrine(orhormonal) systems.[4]These disruptions can cause numerous adverse human health outcomes, including alterations in sperm quality and fertility; abnormalities in sex organs‚endometriosis‚early puberty‚ altered nervous system or immune function; certain cancers; respiratory problems; metabolic issues; diabetes, obesity, or cardiovascular problems; growth, neurological and learning disabilities, and more.[5][6]Found in many household and industrial products, endocrine disruptors "interfere with the synthesis, secretion, transport, binding, action, or elimination of naturalhormonesin the body that are responsible for development, behavior, fertility, and maintenance ofhomeostasis(normal cell metabolism). "[7][8][9]
Any system in the body controlled byhormonescan be derailed by hormone disruptors. Specifically, endocrine disruptors may be associated with the development oflearning disabilities,severeattention deficit disorder,andcognitiveand brain development problems.[10][11][12][13]
There has been controversy over endocrine disruptors, with some groups calling for swift action by regulators to remove them from the market, and regulators and other scientists calling for further study.[14]Some endocrine disruptors have been identified and removed from the market (for example, a drug calleddiethylstilbestrol), but it is uncertain whether some endocrine disruptors on the market actually harm humans and wildlife at the doses to which wildlife and humans are exposed. TheWorld Health Organizationpublished a 2012 report stating that low-level exposures may cause adverse effects in humans.[15]
History
[edit]The termendocrine disruptorwas coined in 1991 at the Wingspread Conference Center in Wisconsin. One of the early papers on the phenomenon was byTheo Colbornin 1993.[16]In this paper, she stated that environmental chemicals disrupt the development of the endocrine system, and that effects of exposure during development are often permanent. Although the endocrine disruption has been disputed by some,[17]work sessions from 1992 to 1999 have generated consensus statements from scientists regarding the hazard from endocrine disruptors, particularly in wildlife and also in humans.[18][19][20][21][22]
The Endocrine Societyreleased a scientific statement outlining mechanisms and effects of endocrine disruptors on "male and female reproduction, breast development and cancer, prostate cancer, neuroendocrinology, thyroid, metabolism and obesity, and cardiovascular endocrinology," and showing how experimental and epidemiological studies converge with human clinical observations "to implicate endocrine disruptive chemicals (EDCs) as a significant concern topublic health."The statement noted that it is difficult to show that endocrine disruptors cause human diseases, and it recommended that theprecautionary principleshould be followed.[23]A concurrent statement expresses policy concerns.[24]
Endocrine disrupting compounds encompass a variety of chemical classes, including drugs, pesticides, compounds used in theplastics industryand in consumer products, industrial by-products and pollutants,heavy metalsand even some naturally produced botanical chemicals. Some are pervasive and widely dispersed in the environment and maybioaccumulate.Some arepersistent organic pollutants(POPs), and can be transported long distances across national boundaries and have been found in virtually all regions of the world, and may even concentrate near the North Pole, due to weather patterns and cold conditions.[25]Others are rapidly degraded in the environment or human body or may be present for only short periods of time.[26]Health effects attributed to endocrine disrupting compounds include a range of reproductive problems (reduced fertility, male and femalereproductive tractabnormalities, and skewed male/femalesex ratios,loss of fetus, menstrual problems[27]); changes in hormone levels; early puberty; brain and behavior problems; impaired immune functions; and various cancers.[28]
One example of the consequences of the exposure of developing animals, including humans, to hormonally active agents is the case of the drugdiethylstilbestrol(DES), anonsteroidalestrogenand not an environmental pollutant. Prior to its ban in the early 1970s, doctors prescribed DES to as many as five million pregnant women to block spontaneous abortion, anoff-label useof this medication prior to 1947. It was discovered after the children went through puberty that DES affected the development of the reproductive system and causedvaginal cancer.The relevance of the DES saga to the risks of exposure to endocrine disruptors is questionable, as the doses involved are much higher in these individuals than in those due to environmental exposures.[29]
Aquaticlife subjected to endocrine disruptors in an urban effluent have experienced decreased levels ofserotoninand increased feminization.[30]
In 2013 theWHOand theUnited Nations Environment Programmereleased a study, the most comprehensive report on EDCs to date, calling for more research to fully understand the associations between EDCs and the risks to health of human and animal life. The team pointed to wide gaps in knowledge and called for more research to obtain a fuller picture of the health and environmental impacts of endocrine disruptors. To improve global knowledge the team has recommended:
- Testing: known EDCs are only the 'tip of the iceberg' and more comprehensive testing methods are required to identify other possible endocrine disruptors, their sources, and routes of exposure.
- Research: more scientific evidence is needed to identify the effects of mixtures of EDCs on humans and wildlife (mainly from industrial by-products) to which humans and wildlife are increasingly exposed.
- Reporting: many sources of EDCs are not known because of insufficient reporting and information on chemicals in products, materials and goods.
- Collaboration: more data sharing between scientists and between countries can fill gaps in data, primarily in developing countries and emerging economies.[31]
Endocrine system
[edit]Endocrine systems are found in most varieties ofanimals.The endocrine system consists ofglandsthat secretehormones,andreceptorsthat detect and react to the hormones.[32]
Hormones travel throughout the body and act as chemical messengers. Hormones interface with cells that contain matching receptors in or on their surfaces. The hormone binds with the receptor, much like a key would fit into a lock. The endocrine system regulates adjustments through slower internal processes, using hormones as messengers. The endocrine system secretes hormones in response to environmental stimuli and to orchestrate developmental and reproductive changes. The adjustments brought on by the endocrine system are biochemical, changing the cell's internal and external chemistry to bring about a long term change in the body.[33]These systems work together to maintain the proper functioning of the body through its entire life cycle.Sex steroidssuch asestrogensandandrogens,as well asthyroidhormones, are subject tofeedbackregulation, which tends to limit the sensitivity of these glands.[34]
Hormones work at very small doses (part per billion ranges). Endocrine disruption can thereby also occur from low-dose exposure to exogenous hormones or hormonally active chemicals such asbisphenol A.These chemicals can bind to receptors for other hormonally mediated processes.[35]Furthermore, sinceendogenoushormones are already present in the body in biologically active concentrations, additional exposure to relatively small amounts ofexogenoushormonally active substances can disrupt the proper functioning of the body's endocrine system. Thus, an endocrine disruptor can elicit adverse effects at much lower doses than a toxicity, acting through a different mechanism.
The timing of exposure is also critical. Most critical stages of development occur in utero, where the fertilized egg divides, rapidly developing every structure of a fully formed baby, including much of the wiring in the brain. Interfering with the hormonal communication in utero can have profound effects both structurally and toward brain development. Depending on the stage of reproductive development, interference with hormonal signaling can result in irreversible effects not seen in adults exposed to the same dose for the same length of time.[36][37][38]Experiments with animals have identified critical developmental time points in utero and days after birth when exposure to chemicals that interfere with or mimic hormones have adverse effects that persist into adulthood.[37][39][40][41]Disruption of thyroid function early in development may be the cause of abnormal sexual development in both males[42]and females[43]early motor development impairment,[44]and learning disabilities.[45]
There are studies of cell cultures, laboratory animals, wildlife, and accidentally exposed humans that show that environmental chemicals cause a wide range of reproductive, developmental, growth, and behavior effects, and so while "endocrine disruption in humans by pollutant chemicals remains largely undemonstrated, the underlying science is sound and the potential for such effects is real."[46]While compounds that produce estrogenic, androgenic,antiandrogenic,andantithyroidactions have been studied, less is known about interactions with other hormones.
The interrelationships between exposures to chemicals and health effects are rather complex. It is hard to definitively link a particular chemical with a specific health effect, and exposed adults may not show any ill effects. But, fetuses and embryos, whose growth and development are highly controlled by the endocrine system, are more vulnerable to exposure and may develop overt or subtle lifelong health or reproductive abnormalities.[47]Prebirth exposure, in some cases, can lead to permanent alterations and adult diseases.[48]
Some in the scientific community are concerned that exposure to endocrine disruptors in the womb or early in life may be associated withneurodevelopmental disordersincluding reduced IQ,ADHD,andautism.[49]Certain cancers and uterine abnormalities in women are associated with exposure todiethylstilbestrol(DES) in the womb due to DES used as a medical treatment.
In a 2005 publication,phthalatesin pregnant women's urine was linked to subtle, but specific, genital changes in their male infants—a shorter, more female-likeanogenital distanceand associated incomplete descent of testes and a smaller scrotum and penis.[50]The science behind this study was questioned by phthalate industry consultants,[51]and back in 2008, there were only five studies of anogenital distance in humans,[52]with one researcher stating, "Whether AGD measures in humans relate to clinically important outcomes, however, remains to be determined, as does its utility as a measure of androgen action in epidemiological studies."[53]Today, it is well-established that AGD is an indicator of fetal androgen exposure, and several studies have found a correlation between AGD and the incidence of prostate cancer.[54][55]
Effects on intrinsic hormones
[edit]Toxicologyresearch shows that some endocrine disruptors target the specific hormone trait that allows one hormone to regulate the production or degradation of intrinsic hormones.[56][57]As endocrine disruptors have the potential to mimic or antagonize natural hormones, these chemicals can exert their effects by acting through interaction withnuclear receptors,thearyl hydrocarbon receptoror membrane bound receptors.[58][59]
U-shaped dose-response curve
[edit]Most toxicants, including endocrine disruptors, have been claimed to follow a U-shapeddose-response curve.[60]This means that very low and very high levels have more effects than mid-level exposure to a toxicant.[61]
Endocrine-disrupting effects have been noted in animals exposed to environmentally relevant levels of some chemicals. For example, a commonflame retardant,BDE-47, affects the reproductive system and thyroid gland of female rats in doses similar to which humans are exposed.[62]
Low concentrations of endocrine disruptors can also have synergistic effects in amphibians, but it is not clear that this is an effect mediated through the endocrine system.[63]
A consensus statement by the Learning and Developmental Disabilities Initiative argued that "The very low-dose effects of endocrine disruptors cannot be predicted from high-dose studies, which contradicts the standard 'dose makes the poison' rule of toxicology. Nontraditional dose-response curves are referred to as non-monotonic dose response curves."[49]
It has been claimed thattamoxifenand somephthalateshave fundamentally different (and harmful) effects on the body at low doses than at high doses.[64]
Routes of exposure
[edit]Food
[edit]Food is a major mechanism by which people are exposed to pollutants. Diet is thought to account for up to 90% of a person'sPCBandDDTbody burden.[65]In a study of 32 different common food products from three grocery stores in Dallas, Texas, fish and other animal products were found to be contaminated withPBDE.[66]Since these compounds are fat-soluble, it is likely they are accumulating from the environment in the fatty tissue of animals eaten by humans. Some suspect fish consumption is a major source of many environmental contaminants. Indeed, both wild and farmed salmon from all over the world have been shown to contain a variety of man-made organic compounds.[67]While pesticides are found in many food products, phthalates can also leech into crops, vegetables and fruits from contaminated soil and greenhouse plastic covers.[68]
Indoor air and household dust
[edit]With the increase in household products containing pollutants and the decrease in the quality of building ventilation, indoor air has become a significant source of pollutant exposure.[69]Residents living in houses with wood floors treated in the 1960s with PCB-based wood finish have a much higher body burden than the general population.[70]A study of indoor house dust and dryer lint of 16 homes found high levels of all 22 different PBDEcongenerstested for in all samples.[71]Recent studies suggest that contaminated house dust, not food, may be the major source of PBDE in the body.[72][73]One study estimated that ingestion of house dust accounts for up to 82% of humans' PBDE body burden.[74]
It has been shown that contaminated house dust is a primary source of lead in young children's bodies.[75]It may be that babies and toddlers ingest more contaminated house dust than the adults they live with, and therefore have much higher levels of pollutants in their systems.
Cosmetics and personal care products
[edit]Consumer goods are another potential source of exposure to endocrine disruptors. An analysis of the composition of 42 household cleaning and personal care products versus 43 "chemical-free" products has been performed. The products contained 55 different chemical compounds: 50 were found in the 42 conventional samples representing 170 product types, while 41 were detected in 43 "chemical-free" samples representing 39 product types.Parabens,a class of chemicals that has been associated with reproductive-tract issues, were detected in seven of the "chemical-free" products, including three sunscreens that did not list parabens on the label. Vinyl products such as shower curtains were found to contain more than 10% by weight of the compoundDEHP,which when present in dust has been associated with asthma and wheezing in children. The risk of exposure to EDCs increases as products, both conventional and "chemical-free", are used in combination. "If a consumer used the alternative surface cleaner, tub and tile cleaner, laundry detergent, bar soap, shampoo and conditioner, facial cleanser and lotion, and toothpaste [he or she] would potentially be exposed to at least 19 compounds: 2 parabens, 3phthalates,MEA,DEA,5alkylphenols,and 7 fragrances. "[76]
An analysis of the endocrine-disrupting chemicals inOld Order Mennonitewomen in mid-pregnancy determined that they have much lower levels in their systems than the general population. Mennonites eat mostly fresh, unprocessed foods, farm without pesticides, and use few or no cosmetics or personal care products. One woman who had reported using hairspray and perfume had high levels of monoethyl phthalate, while the other women all had levels below detection. Three women who reported being in a car or truck within 48 hours of providing a urine sample had higher levels of diethylhexyl phthalate, which is found in polyvinyl chloride and is used in car interiors.[77]
Clothing
[edit]A more recent discussion around exposure to EDCs has been around clothing.
Greenpeacehas reported on endocrine-disrupting chemicals in clothing since 2011. In 2013, Greenpeace found detectable levels ofphthalatesin 33 out of 35 printed articles of clothing from a global sample.[78]A particularly high level ofDEHPwas found in a t-shirt fromPrimarkGermany, and a high level ofDINPwas found in ababyone-piecefromAmerican Apparel.PFCswere commonly found in swimwear and waterproof clothing.NPEswere found in most clothing articles as well.
A study by Greenpeace Germany published in 2014 again found high levels of phthalates in athletic gear.[79]The print of a t-shirt produced in Argentine contained phthalate levels as high as 15%, while a pair of gloves contained 6% phthalates. The study also found high levels ofPFAS,nonoxynolsanddimethylformamidein shoes and boots.
In research published in 2019, Li et al. stated that dermal absorption was the main route forphthalateexposure in infants,[80]including through clothing. It was found that laundering could not remove phthalates completely. Out of the six different types of phthalates that were measured, DEHP andDBPwere found to be particularly present in infant clothing.
Tang et al. published research in 2019 that found all 15 different phthalates that were measured in preschoolers' clothing.[81]Levels were largely independent of country of manufacture though they differed by garment type, fabric composition, and garment color. It was found that "when children wore trousers, long-sleeved shirts, briefs and socks at the same time, the reproductive risks exceeded acceptable level".[81]
In a review of 120 articles from 2014 to 2023 about phthalates in clothing, it was found that whilescreen printingink,[82]vinyl patches andsynthetic leathermay contain 30–60% phthalates,waterproofitems such as infantmattress coversalso contained very high levels of these chemicals.[68]It was also noted that manufacturers work to replace more regulated substances, such as DEHP, with newer ones, that may not yet be as tightly regulated.
Environment
[edit]Additives added toplasticsduring manufacturing may leach into the environment after the plastic item is discarded; additives inmicroplasticsin the ocean leach into ocean water and in plastics inlandfillsmay escape and leach into the soil and then intogroundwater.[83]
Types
[edit]All people are exposed to chemicals with estrogenic effects in their everyday life, because endocrine disrupting chemicals are found in low doses in thousands of products. Chemicals commonly detected in people includeDDT,polychlorinated biphenyls(PCBs),bisphenol A(BPA),polybrominated diphenyl ethers(PBDEs), and a variety ofphthalates.[84]In fact, almost all plastic products, including those advertised as BPA-free, have been found to leach endocrine-disrupting chemicals.[85]In a 2011, study it was found that some BPA-free products released more endocrine-active chemicals than the BPA-containing products.[86][87]Other forms of endocrine disruptors arephytoestrogens(plant hormones).[88]
Xenoestrogens
[edit]Xenoestrogens are a type ofxenohormonethat imitatesestrogen.[89]Synthetic xenoestrogens include widely used industrial compounds, such asPCBs,BPAandphthalates,which have estrogenic effects on a living organism.
Alkylphenols
[edit]Alkylphenols arexenoestrogens.[90]The European Union has implemented sales and use restrictions on certain applications in whichnonylphenolsare used because of their alleged "toxicity, persistence, and the liability to bioaccumulate" but theUnited States Environmental Protections Agency(EPA) has taken a slower approach to make sure that action is based on "sound science".[91]
The long-chain alkylphenols are used extensively as precursors to thedetergents,as additives for fuels andlubricants,polymers, and as components inphenolic resins.These compounds are also used as building block chemicals that are also used in makingfragrances,thermoplasticelastomers,antioxidants,oil field chemicals andfire retardantmaterials. Through the downstream use in making alkylphenolic resins, alkylphenols are also found in tires, adhesives, coatings, carbonless copy paper and high performance rubber products. They have been used in industry for over 40 years.
Certain alkylphenols are degradation products from nonionicdetergents.Nonylphenol is considered to be a low-level endocrine disruptor owing to its tendency to mimic estrogen.[92][93]
Bisphenol A (BPA)
[edit]This sectionneeds morereliable medical referencesforverificationor relies too heavily onprimary sources.(March 2016) |  |

Bisphenol A is commonly found in plastic bottles, plastic food containers, dental materials, and the linings of metalfood and infant formula cans.Another exposure comes from receipt paper commonly used at grocery stores and restaurants, because today the paper is commonly coated with a BPA containing clay for printing purposes.[94]
BPA is a known endocrine disruptor, and numerous studies have found that laboratory animals exposed to low levels of it have elevated rates ofdiabetes,mammaryandprostate cancers,decreased sperm count, reproductive problems, earlypuberty,obesity,and neurological problems.[95][96][97][98]To expand on the reproductive problems faced by women exposed to BPA. Studies in the US have shown that healthy women without any fertility problems found that urinary BPA was unrelated to time of pregnancy despite a shorter luteal phase (second part of the menstrual cycle) being reported.[99][100]Additional studies have been conducted in fertility centers say that BPA exposure is correlation with lower ovarian reserves.[101]To combat this, most women will undergo IVF to help with the poor ovarian stimulation response; seemingly all of them have elevated levels of BPA in the urinary tract.[102]Median conjugation of BPA concentrations were higher in those who did have a miscarriage compared to those who had a live birth.[103]All of these studies show that BPA can have an effect on ovarian functions and the pivotal early part of conception. One study did show racial or ethnic differences as Asian women were found to have an increasedoocytematurity rate, but all of the women had significantly lower concentration of BPA in the study.[104]Early developmental stages appear to be the period of greatest sensitivity to its effects, and some studies have linked prenatal exposure to later physical and neurological difficulties.[105]Regulatory bodies have determined safety levels for humans, but those safety levels are currently being questioned or are under review as a result of new scientific studies.[106][107]A 2011cross-sectional studythat investigated the number of chemicals pregnant women are exposed to in the U.S. found BPA in 96% of women.[108] In 2010 theWorld Health Organizationexpert panel recommended no new regulations limiting or banning the use of bisphenol A, stating that "initiation of public health measures would be premature."[109]
In August 2008, the U.S. FDA issued a draft reassessment, reconfirming their initial opinion that, based on scientific evidence, BPA is safe.[110]However, in October 2008, FDA's advisory Science Board concluded that the Agency's assessment was "flawed" and had not proven the chemical to be safe for formula-fed infants.[111]In January 2010, the FDA issued a report indicating that, due to findings of recent studies that used novel approaches in testing for subtle effects, both the National Toxicology Program at the National Institutes of Health as well as the FDA have some level of concern regarding the possible effects of BPA on the brain and behavior of fetuses, infants and younger children.[112]In 2012 the FDA did ban the use of BPA in baby bottles; however, theEnvironmental Working Groupcalled the ban "purely cosmetic". In a statement they said, "If the agency truly wants to prevent people from being exposed to this toxic chemical associated with a variety of serious and chronic conditions it should ban its use in cans of infant formula, food and beverages." TheNatural Resources Defense Councilcalled the move inadequate saying, the FDA needs to ban BPA from allfood packaging.[113]In a statement a FDA spokesman said the agency's action was not based on safety concerns and that "the agency continues to support the safety of BPA for use in products that hold food."[114]
A program initiated byNIEHS,NTP,and the U.S.Food and Drug Administration(named CLARITY-BPA) found no effect of chronic exposure to BPA on rats[115]and the FDA considers currently authorized uses of BPA to be safe for consumers.[116]
TheEnvironmental Protection Agencyset[when?]a reference dose for BPA at 50 μg/kg/day for mammals, although exposure to doses lower than the reference dose has been shown to affect both male and female reproductive systems.[117]
Bisphenol S (BPS) and bisphenol F (BPF)
[edit]Bisphenol Sand Bisphenol F are analogs of bisphenol A. They are commonly found in thermal receipts, plastics, and household dust.
Traces of BPS have also been found in personal care products.[118]It is more presently being used because of the ban of BPA. BPS is used in place of BPA in BPA-free items. However, BPS and BPF have been shown to be endocrine disruptors as much as BPA.[119][120]
DDT
[edit]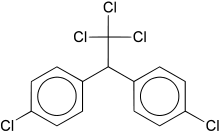
Dichlorodiphenyltrichloroethane (DDT) was first used as a pesticide againstColorado potato beetleson crops beginning in 1936.[121]An increase in the incidence ofmalaria,epidemictyphus,dysentery,andtyphoid feverled to its use against the mosquitoes, lice, and houseflies that carried these diseases. Before World War II,pyrethrum,an extract of a flower from Japan, had been used to control these insects and the diseases they can spread. During World War II, Japan stopped exporting pyrethrum, forcing the search for an alternative. Fearing an epidemic outbreak of typhus, every British and American soldier was issued DDT, who used it to routinely dust beds, tents, and barracks all over the world.
DDT was approved for general, non-military use after the war ended.[121]It became used worldwide to increasemonoculturecrop yields that were threatened by pest infestation, and to reduce the spread of malaria which had a high mortality rate in many parts of the world. Its use for agricultural purposes has since been prohibited by national legislation of most countries, while its use as a control against malaria vectors is permitted, as specifically stated by theStockholm Convention on Persistent Organic Pollutants.[122]
As early as 1946, the harmful effects of DDT on birds, beneficial insects, fish, and marine invertebrates were seen in the environment. The most infamous example of these effects were seen in the eggshells of large predatory birds, which did not develop to be thick enough to support the adult bird sitting on them.[123]Further studies found DDT in high concentrations in carnivores all over the world, the result ofbiomagnificationthrough thefood chain.[124]Twenty years after its widespread use, DDT was found trapped in ice samples taken from Antarctic snow, suggesting wind and water are another means of environmental transport.[125]Recent studies show the historical record of DDT deposition on remote glaciers in the Himalayas.[126]
More than sixty years ago when biologists began to study the effects of DDT on laboratory animals, it was discovered that DDT interfered with reproductive development.[127][128]Recent studies suggest DDT may inhibit the proper development of female reproductive organs that adversely affects reproduction into maturity.[129]Additional studies suggest that a marked decrease in fertility in adult males may be due to DDT exposure.[130]Most recently, it has been suggested that exposure to DDT in utero can increase a child's risk ofchildhood obesity.[131]DDT is still used as anti-malarial insecticide in Africa and parts of Southeast Asia in limited quantities.
Polychlorinated biphenyls
[edit]Polychlorinated biphenyls (PCBs) are a class of chlorinated compounds used as industrial coolants and lubricants. PCBs are created by heating benzene, a byproduct of gasoline refining, with chlorine.[132]They were first manufactured commercially by the Swann Chemical Company in 1927.[133]In 1933, the health effects of direct PCB exposure was seen in those who worked with the chemicals at the manufacturing facility in Alabama. In 1935,Monsantoacquired the company, taking over US production and licensing PCB manufacturing technology internationally.
General Electric was one of the largest US companies to incorporate PCBs into manufactured equipment.[133]Between 1952 and 1977, the New York GE plant had dumped more than 500,000 pounds of PCB waste into the Hudson River. PCBs were first discovered in the environment far from its industrial use by scientists in Sweden studying DDT.[134]
The effects of acute exposure to PCBs were well known within the companies who used Monsanto's PCB formulation who saw the effects on their workers who came into contact with it regularly. Direct skin contact results in a severe acne-like condition calledchloracne.[135]Exposure increases the risk of skin cancer,[136]liver cancer,[137]and brain cancer.[136][138]Monsanto tried for years to downplay the health problems related to PCB exposure in order to continue sales.[139]
The detrimental health effects of PCB exposure to humans became undeniable when two separate incidents of contaminated cooking oilpoisoned thousands of residentsin Japan (Yushō disease, 1968) and Taiwan (Yu-cheng disease, 1979),[140]leading to a worldwide ban on PCB use in 1977. Recent studies show the endocrine interference of certain PCB congeners is toxic to the liver and thyroid,[141]increases childhood obesity in children exposed prenatally,[131]and may increase the risk of developing diabetes.[142][143]
PCBs in the environment may also be related to reproductive and infertility problems in wildlife. In Alaska, it is thought that they may contribute to reproductive defects, infertility and antler malformation in some deer populations. Declines in the populations of otters and sea lions may also be partially due to their exposure to PCBs, the insecticide DDT, other persistent organic pollutants. Bans and restrictions on the use of EDCs have been associated with a reduction in health problems and the recovery of some wildlife populations.[144]
Polybrominated diphenyl ethers
[edit]Polybrominated diphenyl ethers (PBDEs) are a class of compounds found inflame retardantsused in plastic cases of televisions and computers, electronics, carpets, lighting, bedding, clothing, car components, foam cushions and othertextiles.Potential health concern: PBDEs are structurally very similar topolychlorinated biphenyls(PCBs), and have similarneurotoxiceffects.[145]Research has correlatedhalogenated hydrocarbons,such as PCBs, withneurotoxicity.[141]PBDEs are similar in chemical structure to PCBs, and it has been suggested that PBDEs act by the same mechanism as PCBs.[141]
In the 1930s and 1940s, the plastics industry developed technologies to create a variety of plastics with broad applications.[146]OnceWorld War IIbegan, the US military used these new plastic materials to improve weapons, protect equipment, and to replace heavy components in aircraft and vehicles.[146]After WWII, manufacturers saw the potential plastics could have in many industries, and plastics were incorporated into new consumer product designs. Plastics began to replace wood and metal in existing products as well, and today plastics are the most widely used manufacturing materials.[146]
By the 1960s, all homes were wired with electricity and had numerous electrical appliances. Cotton had been the dominant textile used to produce home furnishings,[147]but now home furnishings were composed of mostly synthetic materials. More than 500 billion cigarettes were consumed each year in the 1960s, as compared to less than 3 billion per year in the beginning of the twentieth century.[148]When combined with high-density living, the potential for home fires was higher in the 1960s than it had ever been in the US. By the late 1970s, approximately 6000 people in the US died each year in home fires.[149]
In 1972, in response to this situation, the National Commission on Fire Prevention and Control was created to study the fire problem in the US. In 1973 they published their findings in "America Burning", a 192-page report that made recommendations to increase fire prevention.[150]Most of the recommendations dealt with fire prevention education and improved building engineering, such as the installation of fire sprinklers and smoke detectors. The Commission expected that with the recommendations, a 5% reduction in fire losses could be expected each year, halving the annual losses within 14 years.
Historically, treatments with alum and borax were used to reduce theflammability of fabricand wood, as far back as Roman times.[151]Since it is a non-absorbent material once created, flame retardant chemicals are added to plastic during the polymerization reaction when it is formed. Organic compounds based on halogens like bromine and chlorine are used as the flame retardant additive in plastics, and in fabric based textiles as well.[151]The widespread use of brominatedflame retardantsmay be due to the push from Great Lakes Chemical Corporation (GLCC) to profit from its huge investment in bromine.[152]In 1992, the world market consumed approximately 150,000 tonnes of bromine-based flame retardants, and GLCC produced 30% of the world supply.[151]
PBDEs have the potential to disrupt thyroid hormone balance and contribute to a variety of neurological and developmental deficits, includinglow intelligenceandlearning disabilities.[153][154]Many of the most common PBDE's were banned in theEuropean Unionin 2006.[155]Studies with rodents have suggested that even brief exposure to PBDEs can cause developmental and behavior problems in juvenile rodents[44][156]and exposure interferes with proper thyroid hormone regulation.[157]
Phthalates
[edit]Phthalates are found in some soft toys, flooring, medical equipment, cosmetics and air fresheners. They are of potential health concern because they are known to disrupt the endocrine system of animals, and some research has implicated them in the rise of birth defects of the male reproductive system.[50][158][159]
Although an expert panel has concluded that there is "insufficient evidence" that they can harm the reproductive system of infants,[160]California,[161][162]Washington state,[163]and Europe have banned them from toys. One phthalate,bis(2-ethylhexyl) phthalate(DEHP), used in medical tubing, catheters and blood bags, may harm sexual development in male infants.[158]In 2002, theFood and Drug Administrationreleased a public report which cautioned against exposing male babies to DEHP. Although there are no direct human studies the FDA report states: "Exposure to DEHP has produced a range of adverse effects in laboratory animals, but of greatest concern are effects on the development of the male reproductive system and production of normal sperm in young animals. In view of the available animal data, precautions should be taken to limit the exposure of the developing male to DEHP".[164]Similarly, phthalates may play a causal role in disrupting masculine neurological development when exposed prenatally.[165]
Dibutyl phthalate(DBP) has also disruptedinsulinandglucagonsignaling in animal models.[166]
Perfluorooctanoic acid
[edit]PFOA is a stable chemical that has been used for its grease-, fire-, and water-resistant properties in products such asnon-stickpan coatings, furniture, firefighter equipment, industrial, and other common household items.[167][168]There is evidence to suggest that PFOA is an endocrine disruptor affecting male and female reproductive systems.[168]PFOA delivered to pregnant rats produced male offspring with decreased levels of 3-β and 17-β-hydroxysteroid dehydrogenase,[168]a gene that transcribes for proteins involved in the production of sperm.[169]Adult women have exhibited low progesterone and androstenedione production when exposed to PFOA, leading to menstrual and reproductive health issues.[168] PFOAexerts hormonal effects including alteration of thyroid hormone levels. Blood serum levels of PFOA were associated with an increased time to pregnancy—or "infertility" —in a 2009 study. PFOA exposure is associated with decreased semen quality. PFOA appeared to act as an endocrine disruptor by a potential mechanism on breast maturation in young girls. A C8 Science Panel status report noted an association between exposure in girls and a later onset of puberty.
Other suspected endocrine disruptors
[edit]Some other examples of putative EDCs arepolychlorinated dibenzo-dioxins(PCDDs) and-furans(PCDFs),polycyclic aromatic hydrocarbons(PAHs),phenol derivativesand a number ofpesticides(most prominent beingorganochlorineinsecticides likeendosulfan,kepone(chlordecone) andDDTand its derivatives, the herbicideatrazine,and the fungicidevinclozolin), the contraceptive 17-alphaethinylestradiol,as well as naturally occurringphytoestrogenssuch asgenisteinandmycoestrogenssuch aszearalenone.
The molting in crustaceans is an endocrine-controlled process. In the marine penaeid shrimpLitopenaeus vannamei,exposure toendosulfanresulted increased susceptibility to acute toxicity and increased mortalities in the postmolt stage of the shrimp.[170]
Many sunscreens containoxybenzone,a chemical blocker that provides broad-spectrum UV coverage, yet is subject to a lot of controversy due its potential estrogenic effect in humans.[171]
Tributyltin(TBT) are organotin compounds. For 40 years TBT was used as abiocideinanti-fouling paint,commonly known as bottom paint. TBT has been shown to impact invertebrate and vertebrate development, disrupting the endocrine system, resulting in masculinization, lower survival rates, as well as many health problems in mammals.
Temporal trends of body burden
[edit]Since being banned, the average humanbody burdensof DDT and PCB have been declining.[65][172][173]Since their ban in 1972, the PCB body burden in 2009 is one-hundredth of what it was in the early 1980s. On the other hand, monitoring programs of European breast milk samples have shown that PBDE levels are increasing.[65][173]An analysis of PBDE content in breast milk samples from Europe, Canada, and the US shows that levels are 40 times higher for North American women than for Swedish women, and that levels in North America are doubling every two to six years.[174][175]
It has been discussed that the long-term slow decline in average body temperature observed since the beginning of the industrial revolution[176]may result from disrupted thyroid hormone signalling.[177]
Animal models
[edit]Because endocrine disruptors affect complex metabolic, reproductive, and neuroendocrine systems, they cannot be modeled in in vitro cell based assay. Consequently, animal models are important for access the risk of endocrine disrupting chemicals.[178]
Mice
[edit]There are multiple lines of genetically engineered mice used for lab studies, in this case the lines can be used as population-based genetic foundations. For instance, one population is named Multi-parent and can be a Collaborative Cross (CC) or Diversity Outbred (DO). These mice, while both from the same eight founder strains, have distinct differences.[179][180][181]
The eight founder strains combine strains that are wild-derived (with high genetic diversity) and historically significant biomedical research bred strains. Each genetically differential line is important in EDCs response and also almost all biological processes and traits.[182]
The CC population consists of 83 inbred mouse strains that over many generations in labs came from the eight founder strains. These inbred mice have recombinant genomes that are developed to ensure every strain is equally related, this eradicates population structure and can result in false positives with qualitative trait locus (QTL) mapping.
While DO mice have the identical alleles to the CC mice population, there are two major differences: every individual is unique, allowing for hundreds of individuals to be applied in one mapping study, making DO mice an extremely useful tool for determining genetic relationships; however, DO individuals cannot be reproduced.
Transgenic
[edit]These rodents, mainly mice, have been bred by inserting other genes from another organism to make transgenic lines (thousands of lines) of rodents. The most recent tool used to do this isCRISPR/Cas9, which allows this process to be done more efficiently.[183]
Genes may be manipulated in a particular cell populations if done under the correct conditions.[184]For endocrine-disrupting chemical (EDC) research, these rodents have become an important tool to the point where they can producehumanized mousemodels.[185][186]Additionally, scientists usegene knockoutlines of mice in order to study how certain mechanisms work when impacted by EDCs.[185][186][187][188]Transgenic rodents are an important tool for studies involving the mechanisms that are impacted by EDC but take a long time to produce and are expensive. Additionally, the genes aimed at for knockout are not always successfully targeted resulting in incomplete knockout of a gene or off-target expression.
Social models
[edit]Experiments (gene by environment) with these relatively new rodent models may be able to discover if there are mechanisms that EDCs could impact in the social decline inautism spectrum disorder(ASD) and other behavioral disorders.[189][190]This is becauseprairieandpine volesare socially monogamous, making them a better model for human social behaviors and development in relation to EDCs.[191][192][193][189][194]Additionally the prairie vole genome has been sequenced making it feasible to do the experiments mentioned above.[189][190]These voles can be compared tomontaneandmeadow voleswho are socially promiscuous and solitary, when looking at how different species have various forms of development and social brain structure.[193][189][194]Both monogamous and promiscuous mice species have been used in these types of experiments, for more information studies[195]can expand on this topic.[196][197][195][198]More complex models that have systems that are as close as possible to humans are being looked at. Looking back at more common rodent models, for instance the common ASD mouse, is helpful but does not fully encompass the entirety of a model of human social behaviors.[191][192]
Zebrafish
[edit]The endocrine systems between mammals and fish are similar; because of this,zebrafish(Danio rerio) are a popular lab choice.[199]Zebrafish work well as a model organism, part of which can be attributed to the fact that researchers are able to study them starting from the embryo, as the embryo is nearly transparent.[199]Additionally, zebrafish have DNA sex markers, which allows biologists to individually assign sex to fish; this is particularly important when studying endocrine disruptors as the disruptors can affect how, among other things, the sex organs work. If there is sperm in the ovaries later on through testing, it can then be pinned to the chemical without the chance of it being a genetic abnormality since the sex was determined by the researcher. Besides zebrafish being readily available, and easy to study through different life stages, their genes are similar to humans'—70% of human genes have a zebrafish counterpart and 84% of disease genes in humans have a zebrafish counterpart.[199]Also important is the fact that the vast majority of endocrine disruptors end up in waterways;[199]it is important to know how the disruptors affect fish, which are model organisms as well.
The zebrafish embryos are transparent, relatively small fish (larvae are less than a few millimeters in size).[200]This allows scientists to view the larvae (in vivo) without killing them to study how their organs develop, in particular neurodevelopment and transport of presumed endocrine-disrupting chemicals, thus how their development is impacted by certain chemicals. As a model, they have simple modes of endocrine disruption,[201]along with homologous physiological, sensory, anatomical and signal-transduction mechanisms similar to mammals.[202]Another helpful tool available to scientists is their recorded genome along with multipletransgeniclines accessible for breeding. Zebrafish and mammalian genomes when compared have prominent similarities with about 80% of human genes expressed in the fish. Additionally, zebrafish are also fairly inexpensive to breed and house in a lab partly due to their shorter lifespan and being able to house more of them compared to mammalian models.[203][204][205][200]
Directions of research
[edit]Research on endocrine disruptors is challenged by five complexities requiring special trial designs and sophisticated study protocols:[206]
- Thedissociation of spacemeans that, although disruptors may act by a common pathway viahormone receptors,their impact may also be mediated by effects at the levels oftransport proteins,deiodinases,degradation of hormones or modifiedsetpointsoffeedback loops(i.e.allostatic load).[207]
- Thedissociation of timemay ensue from the fact that unwanted effects may be triggered in a small time window in theembryonalorfetalperiod, but consequences may ensue decades later or even in the generation of grandchildren.[208]
- Thedissociation of substanceresults from additive, multiplicative or more complex interactions of disruptors in combination that yield fundamentally different effects from that of the respective substances alone.[206]
- Thedissociation of doseimplies that dose-effect relationships use to be nonlinear and sometimes evenU-shaped,so that low or medium doses may have stronger effects than high doses.[207]
- Thedissociation of sexreflects the fact that effects may be different depending on whether embryos or fetuses are female or male.[208][209]
Legal approach
[edit]United States
[edit]The multitude of possible endocrine disruptors are technically regulated in the United States by many laws, including theToxic Substances Control Act,theFood Quality Protection Act,[210]theFood, Drug and Cosmetic Act,theClean Water Act,theSafe Drinking Water Act,and theClean Air Act.
TheCongress of the United Stateshas improved the evaluation and regulation process of drugs and other chemicals. The Food Quality Protection Act of 1996 and the Safe Drinking Water Act of 1996 simultaneously provided the first legislative direction requiring theEPAto address endocrine disruption through establishment of a program for screening and testing of chemical substances.
In 1998, the EPA announced the Endocrine Disruptor Screening Program by establishment of a framework for priority setting, screening and testing more than 85,000 chemicals in commerce. While the Food Quality Protection Act only required the EPA to screen pesticides for potential to produce effects similar to estrogens in humans, it also gave the EPA the authority to screen other types of chemicals and endocrine effects.[210]Based on recommendations from an advisory panel, the agency expanded the screening program to include male hormones, the thyroid system, and effects on fish and other wildlife.[210]The basic concept behind the program is that prioritization will be based on existing information about chemical uses, production volume, structure-activity and toxicity. Screening is done by use ofin vitrotest systems (by examining, for instance, if an agent interacts with theestrogen receptoror theandrogen receptor) and via the use of in animal models, such as development of tadpoles and uterine growth in prepubertal rodents. Full-scale testing will examine effects not only in mammals (rats) but also in a number of other species (frogs, fish, birds and invertebrates). Since the theory involves the effects of these substances on a functioning system, animal testing is essential for scientific validity, but has been opposed byanimal rightsgroups. Similarly, proof that these effects occur in humans would require human testing, and such testing also has opposition.
After failing to meet several deadlines to begin testing, the EPA finally announced that they were ready to begin the process of testing dozens of chemical entities that are suspected endocrine disruptors early in 2007, eleven years after the program was announced. When the final structure of the tests was announced there was objection to their design. Critics have charged that the entire process has been compromised by chemical company interference.[211]In 2005, the EPA appointed a panel of experts to conduct an open peer-review of the program and its orientation. Their results found that "the long-term goals and science questions in the EDC program are appropriate",[212]however this study was conducted over a year before the EPA announced the final structure of the screening program. The EPA is still finding it difficult to execute a credible and efficient endocrine testing program.[210]
As of 2016, the EPA had estrogen screening results for 1,800 chemicals.[210]
Europe
[edit]In 2013, a number of pesticides containing endocrine disrupting chemicals were in draft EU criteria to be banned. On 2 May, USTransatlantic Trade and Investment Partnership(TTIP) negotiators insisted the EU drop the criteria. They stated that a risk-based approach should be taken on regulation. Later the same dayCatherine Daywrote to Karl Falkenberg asking for the criteria to be removed.[213]
TheEuropean Commissionhad been to set criteria by December 2013 identifying endocrine-disrupting chemicals (EDCs) in thousands of products—including disinfectants, pesticides and toiletries—that have been linked to cancers, birth defects and development disorders in children. However, the body delayed the process, prompting Sweden to state that it would sue the commission in May 2014—blaming chemical industry lobbying for the disruption.[214]
"This delay is due to the European chemical lobby, which put pressure again on different commissioners. Hormone disrupters are becoming a huge problem. In some places inSwedenwe see double-sexed fish. We have scientific reports on how this affects fertility of young boys and girls, and other serious effects, "Swedish Environment MinisterLena Ektold theAFP,noting thatDenmarkhad also demanded action.[214]
In November 2014, theCopenhagen-basedNordic Council of Ministersreleased its own independent report that estimated the impact of environmental EDCs on male reproductive health, and the resulting cost topublic healthsystems. It concluded that EDCs likely costhealth systemsacross the EU anywhere from 59 million to 1.18 billion Euros a year, noting that even this represented only "a fraction of the endocrine related diseases".[215]
In 2020, the EU published theirChemicals Strategy for Sustainabilitywhich is concerned with agreen transitionof the chemical industry away from xenohormones and otherhazardous chemicals.
Environmental and human body cleanup
[edit]There is evidence that once a pollutant is no longer in use, or once its use is heavily restricted, the humanbody burdenof that pollutant declines. Through the efforts of several large-scale monitoring programs,[84][216]the most prevalent pollutants in the human population are fairly well known. The first step in reducing the body burden of these pollutants is eliminating or phasing out their production.
The second step toward lowering human body burden is awareness of and potentially labeling foods that are likely to contain high amounts of pollutants. This strategy has worked in the past—pregnant and nursing women arecautioned against eating seafoodthat is known to accumulate high levels of mercury.[217]
The most challenging aspect of this problem is discovering how to eliminate these compounds from the environment and where to focus remediation efforts. Even pollutants no longer in production persist in the environment and bio-accumulate in the food chain. An understanding of how these chemicals, once in the environment, move through ecosystems, is essential to designing ways to isolate and remove them. Global efforts have been made to label the most common POPs routinely found in the environment through usage of chemicals like insecticides. The twelve main POPs have been evaluated and placed in a demographic so as to streamline the information around the general population. Such facilitation has allowed nations around the world to effectively work on the testing and reduction of the usage of these chemicals. With an effort to reduce the presence of such chemicals in the environment, they can reduce the leaching of POPs into food sources which contaminate the animals commercially fed to the U.S. population.[218]
Many persistent organic compounds, PCB, DDT and PBDE included, accumulate in river and marine sediments. Several processes are currently being used by the EPA to clean up heavily polluted areas, as outlined in theirGreen Remediationprogram.[219]
Naturally occurring microbes that degrade PCB congeners to remediate contaminated areas are utilized.[220]
There are many success stories of cleanup efforts of large heavily contaminatedSuperfundsites. A 10-acre (40,000 m2) landfill in Austin, Texas, contaminated with illegally dumpedVOCswas restored in a year to a wetland and educational park.[221]
A US uranium enrichment site that was contaminated with uranium and PCBs was cleaned up with high tech equipment used to find the pollutants within the soil.[222]The soil and water at a polluted wetlands site were cleaned of VOCs, PCBs and lead, native plants were installed asbiological filters,and a community program was implemented to ensure ongoing monitoring of pollutant concentrations in the area.[223]These case studies are encouraging due to the short amount of time needed to remediate the site and the high level of success achieved.
Studies suggest that bisphenol A,[224]certain PCBs,[225]and phthalate compounds[226]are preferentially eliminated from the human body through sweat. Although some pollutants like bisphenol A (BPA) are preferentially eliminated from the human body through sweat, recent scientific advances have been made to increase the rate of elimination of pollutants from the human body. For example, BPA removal techniques have been proposed that use enzymes such aslaccaseandperoxidaseto degrade BPA into less harmful compounds. Another technique for BPA removal is the use of highly reactive radicals for degradation.[227]
Economic effects
[edit]Human exposure may cause some health effects, such as lower IQ and adult obesity. These effects may lead to lost productivity, disability, or premature death in some people. One source estimated that, within theEuropean Union,this economic effect might have about twice theeconomic impactas the effects caused by mercury and lead contamination.[228]
The socio-economic burden of EDC-associated health effects for the European Union was estimated based on currently available literature in 2016 and considering the uncertainties with respect to causality with EDCs and corresponding health-related costs to be in the range of €46 billion to €288 billion per year.[229]
See also
[edit]- Antiandrogens in the environment
- Theo Colborn
- Endocrine system
- Environmental hormones
- Hormone
- Obesogen
- Pollutant-induced abnormal behaviour
- Precautionary principle
- Stink!
- John Sumpter
- Xenoestrogen
References
[edit]- ^Krimsky S (December 2001). "An epistemological inquiry into the endocrine disruptor thesis".Annals of the New York Academy of Sciences.948(1): 130–142.Bibcode:2001NYASA.948..130K.doi:10.1111/j.1749-6632.2001.tb03994.x.PMID11795392.S2CID41532171.
- ^Diamanti-Kandarakis E, Bourguignon JP, Giudice LC, Hauser R, Prins GS, Soto AM, et al. (June 2009)."Endocrine-disrupting chemicals: an Endocrine Society scientific statement"(PDF).Endocrine Reviews.30(4): 293–342.doi:10.1210/er.2009-0002.PMC2726844.PMID19502515.Archived fromthe original(PDF)on 29 September 2009.Retrieved26 September2009.
- ^"Endocrine Disrupting Compounds".National Institutes of Health · U.S. Department of Health and Human Services. Archived fromthe originalon 24 September 2009.
- ^Casals-Casas C, Desvergne B (17 March 2011). "Endocrine disruptors: from endocrine to metabolic disruption".Annual Review of Physiology.73(1): 135–162.doi:10.1146/annurev-physiol-012110-142200.PMID21054169.
- ^"Endocrine-Disrupting Chemicals (EDCs)".www.endocrine.org.24 January 2022.Retrieved20 September2023.
- ^Staff (5 June 2013)."Endocrine Disruptors".NIEHS.
- ^Vandenberg LN, Colborn T, Hayes TB, Heindel JJ, Jacobs DR, Lee DH, et al. (June 2012)."Hormones and endocrine-disrupting chemicals: low-dose effects and nonmonotonic dose responses".Endocrine Reviews.33(3): 378–455.doi:10.1210/er.2011-1050.PMC3365860.PMID22419778.
- ^Crisp TM, Clegg ED, Cooper RL, Wood WP, Anderson DG, Baetcke KP, et al. (February 1998)."Environmental endocrine disruption: an effects assessment and analysis".Environmental Health Perspectives.106.106(Suppl 1): 11–56.doi:10.2307/3433911.JSTOR3433911.PMC1533291.PMID9539004.
- ^Huang AC, Nelson C, Elliott JE, Guertin DA, Ritland C, Drouillard K, et al. (July 2018)."River otters (Lontra canadensis)" trapped "in a coastal environment contaminated with persistent organic pollutants: Demographic and physiological consequences".Environmental Pollution.238:306–316.Bibcode:2018EPoll.238..306H.doi:10.1016/j.envpol.2018.03.035.PMID29573713.
- ^Eskenazi B, Chevrier J, Rauch SA, Kogut K, Harley KG, Johnson C, et al. (February 2013)."In utero and childhood polybrominated diphenyl ether (PBDE) exposures and neurodevelopment in the CHAMACOS study".Environmental Health Perspectives.121(2): 257–62.doi:10.1289/ehp.1205597.PMC3569691.PMID23154064.
- ^Jurewicz J, Hanke W (June 2011)."Exposure to phthalates: reproductive outcome and children health. A review of epidemiological studies".International Journal of Occupational Medicine and Environmental Health.24(2): 115–41.doi:10.2478/s13382-011-0022-2.PMID21594692.
- ^Bornehag CG, Engdahl E, Unenge Hallerbäck M, Wikström S, Lindh C, Rüegg J, et al. (May 2021)."Prenatal exposure to bisphenols and cognitive function in children at 7 years of age in the Swedish SELMA study".Environment International.150:106433.Bibcode:2021EnInt.15006433B.doi:10.1016/j.envint.2021.106433.PMID33637302.S2CID232064637.
- ^Repouskou A, Papadopoulou AK, Panagiotidou E, Trichas P, Lindh C, Bergman Å, et al. (June 2020)."Long term transcriptional and behavioral effects in mice developmentally exposed to a mixture of endocrine disruptors associated with delayed human neurodevelopment".Scientific Reports.10(1): 9367.Bibcode:2020NatSR..10.9367R.doi:10.1038/s41598-020-66379-x.PMC7283331.PMID32518293.
- ^Lupu D, Andersson P, Bornehag CG, Demeneix B, Fritsche E, Gennings C, et al. (June 2020)."The ENDpoiNTs Project: Novel Testing Strategies for Endocrine Disruptors Linked to Developmental Neurotoxicity".International Journal of Molecular Sciences.21(11): 3978.doi:10.3390/ijms21113978.PMC7312023.PMID32492937.
- ^"State of the science of endocrine disrupting chemicals".World Health Organization. 2012.Retrieved20 October2023.
- ^Colborn T, vom Saal FS, Soto AM (October 1993)."Developmental effects of endocrine-disrupting chemicals in wildlife and humans".Environ. Health Perspect.101(5): 378–84.doi:10.2307/3431890.JSTOR3431890.PMC1519860.PMID8080506.
- ^Grady D (6 September 2010)."In Feast of Data on BPA Plastic, No Final Answer".The New York Times.
A fierce debate has resulted, with some dismissing the whole idea of endocrine disruptors.
- ^Bern HA, Blair P, Brasseur S, Colborn T, Cunha GR, Davis W, et al. (1992)."Statement from the Work Session on Chemically-Induced Alterations in Sexual Development: The Wildlife/Human Connection"(PDF).In Clement C, Colborn T (eds.).Chemically-induced alterations in sexual and functional development-- the wildlife/human connection.Princeton, N.J: Princeton Scientific Pub. Co. pp. 1–8.ISBN978-0-911131-35-2.Archived fromthe original(PDF)on 26 July 2011.Retrieved26 September2010.
- ^Bantle J, Bowerman WW IV, Carey C, Colborn T, Deguise S, Dodson S, et al. (May 1995)."Statement from the Work Session on Environmentally induced Alterations in Development: A Focus on Wildlife".Environmental Health Perspectives.103(Suppl 4): 3–5.doi:10.2307/3432404.JSTOR3432404.PMC1519268.PMID17539108.
- ^Benson WH, Bern HA, Bue B, Colborn T, Cook P, Davis WP, et al. (1997)."Statement from the work session on chemically induced alterations in functional development and reproduction of fishes".In Rolland RM, Gilbertson M, Peterson RE (eds.).Chemically Induced Alterations in Functional Development and Reproduction of Fishes.Society of Environmental Toxicology & Chemist. pp.3–8.ISBN978-1-880611-19-7.
- ^Alleva E, Brock J, Brouwer A, Colborn T, Fossi MC, Gray E, et al. (1998). "Statement from the work session on environmental endocrine-disrupting chemicals: neural, endocrine, and behavioral effects".Toxicology and Industrial Health.14(1–2): 1–8.Bibcode:1998ToxIH..14....1..doi:10.1177/074823379801400103.PMID9460166.S2CID45902764.
- ^Brock J, Colborn T, Cooper R, Craine DA, Dodson SF, Garry VF, et al. (1999). "Statement from the Work Session on Health Effects of Contemporary-Use Pesticides: the Wildlife / Human Connection".Toxicol Ind Health.15(1–2): 1–5.Bibcode:1999ToxIH..15....1..doi:10.1191/074823399678846547.
- ^Diamanti-Kandarakis E, Bourguignon JP, Giudice LC, Hauser R, Prins GS, Soto AM, et al. (June 2009)."Endocrine-disrupting chemicals: an Endocrine Society scientific statement".Endocrine Reviews.30(4): 293–342.doi:10.1210/er.2009-0002.PMC2726844.PMID19502515.
- ^"Position statement: Endocrine-disrupting chemicals"(PDF).Endocrine News.34(8): 24–27. 2009. Archived fromthe original(PDF)on 30 October 2010.
- ^Visser MJ."Cold, Clear, and Deadly".Retrieved14 April2012.
- ^Damstra T, Barlow S, Bergman A, Kavlock R, Van der Kraak G (2002)."REPIDISCA-Global assessment of the state-of-the-science of endocrine disruptors".International programme on chemical safety, World Health Organization.Retrieved14 March2009.
- ^Harrison PT, Humfrey CD, Litchfield M, Peakall D, Shuker LK (1995)."Environmental oestrogens: consequences to human health and wildlife"(PDF).IEH assessment.Medical Research Council, Institute for Environment and Health. Archived fromthe original(PDF)on 28 September 2011.Retrieved14 March2009.
- ^"EDC Human Effects".e.hormone.Center for Bioenvironmental Research at Tulane and Xavier Universities.Retrieved14 March2009.
- ^Golden RJ, Noller KL, Titus-Ernstoff L, Kaufman RH, Mittendorf R, Stillman R, et al. (March 1998). "Environmental endocrine modulators and human health: an assessment of the biological evidence".Crit. Rev. Toxicol.28(2): 109–227.doi:10.1080/10408449891344191.PMID9557209.
- ^Willis IC (2007).Progress in Environmental Research.New York: Nova Publishers. p. 176.ISBN978-1-60021-618-3.
- ^"State of the science of endocrine disrupting chemicals – 2012".World Health Organization. 2013. Archived fromthe originalon 23 February 2013.Retrieved6 April2015.
- ^"Anatomy of the Endocrine System".Johns Hopkins Medicine.19 November 2019.Retrieved11 April2023.
- ^"Hormonal (endocrine) system".Better Health Channel.The Department of Health, Victorian Government.Retrieved11 April2023.
- ^Kim YJ, Tamadon A, Park HT, Kim H, Ku SY (September 2016)."The role of sex steroid hormones in the pathophysiology and treatment of sarcopenia".Osteoporosis and Sarcopenia.2(3): 140–155.doi:10.1016/j.afos.2016.06.002.PMC6372754.PMID30775480.
- ^"Bisphenol A Overview".Environment California. Archived fromthe originalon 22 April 2011.
- ^Guo YL, Lambert GH, Hsu CC (September 1995)."Growth abnormalities in the population exposed in utero and early postnatally to polychlorinated biphenyls and dibenzofurans".Environ. Health Perspect.103(Suppl 6): 117–22.doi:10.2307/3432359.JSTOR3432359.PMC1518940.PMID8549457.
- ^abBigsby R, Chapin RE, Daston GP, Davis BJ, Gorski J, Gray LE, et al. (August 1999)."Evaluating the effects of endocrine disruptors on endocrine function during development".Environ. Health Perspect.107(Suppl 4): 613–8.doi:10.2307/3434553.JSTOR3434553.PMC1567510.PMID10421771.
- ^Castro DJ, Löhr CV, Fischer KA, Pereira CB, Williams DE (December 2008)."Lymphoma and lung cancer in offspring born to pregnant mice dosed with dibenzo[a, l]pyrene: the importance of in utero vs. lactational exposure".Toxicol. Appl. Pharmacol.233(3): 454–8.Bibcode:2008ToxAP.233..454C.doi:10.1016/j.taap.2008.09.009.PMC2729560.PMID18848954.
- ^Eriksson P, Lundkvist U, Fredriksson A (1991). "Neonatal exposure to 3,3′,4,4′-tetrachlorobiphenyl: changes in spontaneous behaviour and cholinergic muscarinic receptors in the adult mouse".Toxicology.69(1): 27–34.doi:10.1016/0300-483X(91)90150-Y.PMID1926153.
- ^Recabarren SE, Rojas-García PP, Recabarren MP, Alfaro VH, Smith R, Padmanabhan V, et al. (December 2008)."Prenatal testosterone excess reduces sperm count and motility".Endocrinology.149(12): 6444–8.doi:10.1210/en.2008-0785.PMID18669598.
- ^Szabo DT, Richardson VM, Ross DG, Diliberto JJ, Kodavanti PR, Birnbaum LS (January 2009)."Effects of perinatal PBDE exposure on hepatic phase I, phase II, phase III, and deiodinase 1 gene expression involved in thyroid hormone metabolism in male rat pups".Toxicol. Sci.107(1): 27–39.doi:10.1093/toxsci/kfn230.PMC2638650.PMID18978342.
- ^Lilienthal H, Hack A, Roth-Härer A, Grande SW, Talsness CE (February 2006)."Effects of developmental exposure to 2,2′,4,4′,5-pentabromodiphenyl ether (PBDE-99) on sex steroids, sexual development, and sexually dimorphic behavior in rats".Environmental Health Perspectives.114(2): 194–201.doi:10.1289/ehp.8391.PMC1367831.PMID16451854.
- ^Talsness CE, Shakibaei M, Kuriyama SN, Grande SW, Sterner-Kock A, Schnitker P, et al. (July 2005). "Ultrastructural changes observed in rat ovaries following in utero and lactational exposure to low doses of a polybrominated flame retardant".Toxicol. Lett.157(3): 189–202.doi:10.1016/j.toxlet.2005.02.001.PMID15917144.
- ^abEriksson P, Viberg H, Jakobsson E, Orn U, Fredriksson A (May 2002)."A brominated flame retardant, 2,2′,4,4′,5-pentabromodiphenyl ether: uptake, retention, and induction of neurobehavioral alterations in mice during a critical phase of neonatal brain development".Toxicol. Sci.67(1): 98–103.doi:10.1093/toxsci/67.1.98.PMID11961221.
- ^Viberg H, Johansson N, Fredriksson A, Eriksson J, Marsh G, Eriksson P (July 2006)."Neonatal exposure to higher brominated diphenyl ethers, hepta-, octa-, or nonabromodiphenyl ether, impairs spontaneous behavior and learning and memory functions of adult mice".Toxicol. Sci.92(1): 211–8.doi:10.1093/toxsci/kfj196.PMID16611620.
- ^Rogan WJ, Ragan NB (July 2003)."Evidence of effects of environmental chemicals on the endocrine system in children".Pediatrics.112(1 Pt 2): 247–52.doi:10.1542/peds.112.S1.247.PMID12837917.S2CID13058233.
- ^Bern HA (November 1992). "The development of the role of hormones in development—a double remembrance".Endocrinology.131(5): 2037–8.doi:10.1210/endo.131.5.1425407.PMID1425407.
- ^Colborn T, Carroll LE (2007). "Pesticides, sexual development, reproduction, and fertility: current perspective and future".Human and Ecological Risk Assessment.13(5): 1078–1110.doi:10.1080/10807030701506405.S2CID34600913.
- ^abCollaborative on Health, the Environment's Learning, Developmental Disabilities Initiative (1 July 2008)."Scientific Consensus Statement on Environmental Agents Associated with Neurodevelopmental Disorders"(PDF).Institute for Children's Environmental Health.Retrieved14 March2009.
- ^abSwan SH, Main KM, Liu F, Stewart SL, Kruse RL, Calafat AM, et al. (August 2005)."Decrease in anogenital distance among male infants with prenatal phthalate exposure".Environmental Health Perspectives.113(8): 1056–61.doi:10.1289/ehp.8100.PMC1280349.PMID16079079.
- ^McEwen GN, Renner G (January 2006)."Validity of anogenital distance as a marker of in utero phthalate exposure".Environmental Health Perspectives.114(1): A19–20, author reply A20–1.doi:10.1289/ehp.114-a19b.PMC1332693.PMID16393642.
- ^Postellon DC (June 2008). "Baby care products".Pediatrics.121(6): 1292, author reply 1292–3.doi:10.1542/peds.2008-0401.PMID18519505.S2CID27956545.
- ^Romano-Riquer SP, Hernández-Avila M,Gladen BC,Cupul-Uicab LA, Longnecker MP (May 2007)."Reliability and determinants of anogenital distance and penis dimensions in male newborns from Chiapas, Mexico".Paediatr Perinat Epidemiol.21(3): 219–28.doi:10.1111/j.1365-3016.2007.00810.x.PMC3653615.PMID17439530.
- ^Maldonado-Cárceles AB, Sánchez-Rodríguez C, Vera-Porras EM, Árense-Gonzalo JJ, Oñate-Celdrán J, Samper-Mateo P, et al. (March 2017). "Anogenital Distance, a Biomarker of Prenatal Androgen Exposure Is Associated With Prostate Cancer Severity".The Prostate.77(4): 406–411.doi:10.1002/pros.23279.PMID27862129.
- ^Marín-Martínez FM, Arense-Gonzalo JJ, Artes MA, Bobadilla Romero ER, García Porcel VJ, Alcon Cerro P, et al. (November 2023). "Anogenital distance, a biomarker of fetal androgen exposure and the risk of prostate cancer: A case-control study".Urologia.90(4): 715–719.doi:10.1177/03915603231192736.PMID37606191.
- ^Harold Z (2011).Human Toxicology of Chemical Mixtures(2nd ed.). Elsevier.ISBN978-1-4377-3463-8.
- ^Yu MH, Tsunoda H, Tsunoda M (2016).Environmental Toxicology: Biological and Health Effects of Pollutants(Third ed.). CRC Press.ISBN978-1-4398-4038-2.
- ^Toporova L, Balaguer P (February 2020)."Nuclear receptors are the major targets of endocrine disrupting chemicals"(PDF).Molecular and Cellular Endocrinology.502:110665.doi:10.1016/j.mce.2019.110665.PMID31760044.S2CID209493576.
- ^Balaguer P, Delfosse V, Grimaldi M, Bourguet W (1 September 2017)."Structural and functional evidences for the interactions between nuclear hormone receptors and endocrine disruptors at low doses".Comptes Rendus Biologies.Endocrine disruptors / Les perturbateurs endocriniens.340(9–10): 414–420.doi:10.1016/j.crvi.2017.08.002.PMID29126514.
- ^Calabrese EJ, Baldwin LA (February 2003). "Toxicology rethinks its central belief".Nature.421(6924): 691–2.Bibcode:2003Natur.421..691C.doi:10.1038/421691a.PMID12610596.S2CID4419048.
- ^Steeger T, Tietge J (29 May 2003).White Paper on Potential Developmental Effects of Atrazine on Amphibians(PDF)(Report). Archived fromthe original(PDF)on 11 July 2004.
- ^Talsness CE, Kuriyama SN, Sterner-Kock A, Schnitker P, Grande SW, Shakibaei M, et al. (March 2008)."In utero and lactational exposures to low doses of polybrominated diphenyl ether-47 alter the reproductive system and thyroid gland of female rat offspring".Environmental Health Perspectives.116(3): 308–14.doi:10.1289/ehp.10536.PMC2265047.PMID18335096.
- ^Hayes TB, Case P, Chui S, Chung D, Haeffele C, Haston K, et al. (April 2006)."Pesticide mixtures, endocrine disruption, and amphibian declines: are we underestimating the impact?".Environmental Health Perspectives.114(S–1): 40–50.doi:10.1289/ehp.8051.PMC1874187.PMID16818245.
- ^Curwood S, Young J (4 September 2009)."Low Dose Makes the Poison".Living on Earth.
- ^abcFürst P (October 2006). "Dioxins, polychlorinated biphenyls and other organohalogen compounds in human milk. Levels, correlations, trends and exposure through breastfeeding".Mol Nutr Food Res.50(10): 922–33.doi:10.1002/mnfr.200600008.PMID17009213.
- ^Schecter A, Päpke O, Tung KC, Staskal D, Birnbaum L (October 2004). "Polybrominated diphenyl ethers contamination of United States food".Environ. Sci. Technol.38(20): 5306–11.Bibcode:2004EnST...38.5306S.doi:10.1021/es0490830.PMID15543730.
- ^Hites RA, Foran JA, Carpenter DO, Hamilton MC, Knuth BA, Schwager SJ (January 2004). "Global assessment of organic contaminants in farmed salmon".Science.303(5655): 226–9.Bibcode:2004Sci...303..226H.doi:10.1126/science.1091447.PMID14716013.S2CID24058620.
- ^abAldegunde-Louzao N, Lolo-Aira M, Herrero-Latorre C (June 2024)."Phthalate esters in clothing: A review".Environmental Toxicology and Pharmacology.108:104457.Bibcode:2024EnvTP.10804457A.doi:10.1016/j.etap.2024.104457.PMID38677495.
- ^Weschler CJ (2009). "Changes in indoor pollutants since the 1950s".Atmospheric Environment.43(1): 153–169.Bibcode:2009AtmEn..43..153W.doi:10.1016/j.atmosenv.2008.09.044.
- ^Rudel RA, Seryak LM, Brody JG (2008)."PCB-containing wood floor finish is a likely source of elevated PCBs in residents' blood, household air and dust: a case study of exposure".Environ Health.7(1): 2.Bibcode:2008EnvHe...7....2R.doi:10.1186/1476-069X-7-2.PMC2267460.PMID18201376.
- ^Stapleton HM, Dodder NG, Offenberg JH, Schantz MM, Wise SA (February 2005). "Polybrominated diphenyl ethers in house dust and clothes dryer lint".Environ. Sci. Technol.39(4): 925–31.Bibcode:2005EnST...39..925S.doi:10.1021/es0486824.PMID15773463.
- ^Anderson HA, Imm P, Knobeloch L, Turyk M, Mathew J, Buelow C, et al. (September 2008). "Polybrominated diphenyl ethers (PBDE) in serum: findings from a US cohort of consumers of sport-caught fish".Chemosphere.73(2): 187–94.Bibcode:2008Chmsp..73..187A.doi:10.1016/j.chemosphere.2008.05.052.PMID18599108.
- ^Morland KB, Landrigan PJ, Sjödin A, Gobeille AK, Jones RS, McGahee EE, et al. (December 2005)."Body burdens of polybrominated diphenyl ethers among urban anglers".Environmental Health Perspectives.113(12): 1689–92.doi:10.1289/ehp.8138.PMC1314906.PMID16330348.
- ^Lorber M (January 2008)."Exposure of Americans to polybrominated diphenyl ethers".J Expo Sci Environ Epidemiol.18(1): 2–19.doi:10.1038/sj.jes.7500572.PMID17426733.
- ^Charney E, Sayre J, Coulter M (February 1980). "Increased lead absorption in inner city children: where does the lead come from?".Pediatrics.65(2): 226–31.doi:10.1542/peds.65.2.226.PMID7354967.
- ^Dodson RE, Nishioka M, Standley LJ, Perovich LJ, Brody JG, Rudel RA (March 2012)."Endocrine Disruptors and Asthma-Associated Chemicals in Consumer Products".Environmental Health Perspectives.120(7): 935–943.doi:10.1289/ehp.1104052.PMC3404651.PMID22398195.
- Lay summary in:Olver C (5 April 2012)."Endocrine disruptors and asthma-associated chemicals in consumer products".Journalist's Resource.
- ^Martina CA, Weiss B, Swan SH (June 2012)."Lifestyle behaviors associated with exposures to endocrine disruptors".Neurotoxicology.33(6): 1427–1433.Bibcode:2012NeuTx..33.1427M.doi:10.1016/j.neuro.2012.05.016.PMC3641683.PMID22739065.
- Lay summary in:"Simpler lifestyle found to reduce exposure to endocrine disrupting chemicals".Science Daily(Press release). 26 June 2012.
- ^Brigden K, Hetherington S, Wang M, Santillo D, Johnston P (June 2013)."Hazardous chemicals in branded textile products on sale in 25 countries/regions during 2013"(PDF).Greenpeace Research Laboratories (published December 2013).
- ^Cobbing M, Brodde K (May 2014)."A Red Card for sportswear brands"(PDF).Greenpeace e.V.
- ^Li HL, Ma WL, Liu LY, Zhang Z, Sverko E, Zhang ZF, et al. (September 2019). "Phthalates in infant cotton clothing: Occurrence and implications for human exposure".The Science of the Total Environment.683:109–115.Bibcode:2019ScTEn.683..109L.doi:10.1016/j.scitotenv.2019.05.132.PMID31129321.
- ^abTang Z, Chai M, Wang Y, Cheng J (April 2020). "Phthalates in preschool children's clothing manufactured in seven Asian countries: Occurrence, profiles and potential health risks".Journal of Hazardous Materials.387:121681.Bibcode:2020JHzM..38721681T.doi:10.1016/j.jhazmat.2019.121681.PMID31757725.
- ^Mohapatra P, Gaonkar O (2021).An Overview of Chemicals in Textiles.Toxics Link(Report). New Delhi, India. p. 41.
- ^Teuten EL, Saquing JM, Knappe DR, Barlaz MA, Jonsson S, Björn A, et al. (2009)."Transport and release of chemicals from plastics to the environment and to wildlife".Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences.364(1526): 2027–45.doi:10.1098/rstb.2008.0284.PMC2873017.PMID19528054.
- ^abFourth national report on human exposure to environmental chemicals (Report). National Center for Environmental Health. 2021.doi:10.15620/cdc:105345.S2CID241050234.
- ^Yang CZ, Yaniger SI, Jordan VC, Klein DJ, Bittner GD (July 2011)."Most plastic products release estrogenic chemicals: a potential health problem that can be solved".Environmental Health Perspectives.119(7): 989–96.doi:10.1289/ehp.1003220.PMC3222987.PMID21367689.
- ^"Study: Most plastic products trigger estrogen effect".USA Today.7 March 2011.
- ^"Study: Even" BPA-Free "Plastics Leach Endrocrine-Disrupting Chemicals".Time.8 March 2011.
- ^"Endocrine Disruptors"(PDF).National Institute of Environmental Health Sciences. May 2010.Retrieved1 January2014.
- ^Watson CS, Bulayeva NN, Wozniak AL, Alyea RA (February 2007)."Xenoestrogens are potent activators of nongenomic estrogenic responses".Steroids.72(2): 124–134.doi:10.1016/j.steroids.2006.11.002.PMC1862644.PMID17174995.
- ^Kochukov MY, Jeng YJ, Watson CS (May 2009)."Alkylphenol xenoestrogens with varying carbon chain lengths differentially and potently activate signaling and functional responses in GH3/B6/F10 somatomammotropes".Environmental Health Perspectives.117(5): 723–30.doi:10.1289/ehp.0800182.PMC2685833.PMID19479013.
- ^Renner R (1997). "European Bans on Surfactant Trigger Transatlantic Debate".Environmental Science & Technology.31(7): 316A–320A.Bibcode:1997EnST...31..316R.doi:10.1021/es972366q.PMID21650741.
- ^Soares A, Guieysse B, Jefferson B, Cartmell E, Lester JN (October 2008). "Nonylphenol in the environment: a critical review on occurrence, fate, toxicity and treatment in wastewaters".Environ Int.34(7): 1033–49.Bibcode:2008EnInt..34.1033S.doi:10.1016/j.envint.2008.01.004.PMID18282600.
- ^"Statewide Endocrine Disrupting Compound Monitoring Study, 2007 – 2008"(PDF).Minnesota Pollution Control Agency.
- ^"Receipts a large — and largely ignored — source of BPA".Science News.178(5): 5. August 2010.
- ^Gore AC(2007).Endocrine-Disrupting Chemicals: From Basic Research to Clinical Practice (Contemporary Endocrinology).Totowa, NJ: Humana Press.ISBN978-1-58829-830-0.
- ^O'Connor JC, Chapin RE (2003)."Critical evaluation of observed adverse effects of endocrine active substances on reproduction and development, the immune system, and the nervous system".Pure Appl. Chem.75(11–12): 2099–2123.doi:10.1351/pac200375112099.S2CID97899046.
- ^Okada H, Tokunaga T, Liu X, Takayanagi S, Matsushima A, Shimohigashi Y (January 2008)."Direct evidence revealing structural elements essential for the high binding ability of bisphenol A to human estrogen-related receptor-gamma".Environmental Health Perspectives.116(1): 32–38.doi:10.1289/ehp.10587.PMC2199305.PMID18197296.
- ^vom Saal FS, Myers JP (September 2008). "Bisphenol A and risk of metabolic disorders".JAMA.300(11): 1353–1355.doi:10.1001/jama.300.11.1353.PMID18799451.
- ^Buck Louis GM, Sundaram R, Sweeney AM, Schisterman EF, Maisog J, Kannan K (May 2014)."Urinary bisphenol A, phthalates, and couple fecundity: the Longitudinal Investigation of Fertility and the Environment (LIFE) Study".Fertility and Sterility.101(5): 1359–1366.doi:10.1016/j.fertnstert.2014.01.022.PMC4008721.PMID24534276.
- ^Jukic AM, Calafat AM, McConnaughey DR, Longnecker MP, Hoppin JA, Weinberg CR, et al. (March 2016)."Urinary Concentrations of Phthalate Metabolites and Bisphenol A and Associations with Follicular-Phase Length, Luteal-Phase Length, Fecundability, and Early Pregnancy Loss".Environmental Health Perspectives.124(3): 321–328.doi:10.1289/ehp.1408164.PMC4786975.PMID26161573.
- ^Souter I, Smith KW, Dimitriadis I, Ehrlich S, Williams PL, Calafat AM, et al. (December 2013)."The association of bisphenol-A urinary concentrations with antral follicle counts and other measures of ovarian reserve in women undergoing infertility treatments".Reproductive Toxicology.42:224–231.Bibcode:2013RepTx..42..224S.doi:10.1016/j.reprotox.2013.09.008.PMC4383527.PMID24100206.
- ^Mok-Lin E, Ehrlich S, Williams PL, Petrozza J, Wright DL, Calafat AM, et al. (April 2010)."Urinary bisphenol A concentrations and ovarian response among women undergoing IVF".International Journal of Andrology.33(2): 385–393.doi:10.1111/j.1365-2605.2009.01014.x.PMC3089904.PMID20002217.
- ^Lathi RB, Liebert CA, Brookfield KF, Taylor JA, vom Saal FS, Fujimoto VY, et al. (July 2014)."Conjugated bisphenol A in maternal serum in relation to miscarriage risk".Fertility and Sterility.102(1): 123–128.doi:10.1016/j.fertnstert.2014.03.024.PMC4711263.PMID24746738.
- ^Fujimoto VY, Kim D, vom Saal FS, Lamb JD, Taylor JA, Bloom MS (April 2011)."Serum unconjugated bisphenol A concentrations in women may adversely influence oocyte quality during in vitro fertilization".Fertility and Sterility.95(5): 1816–1819.doi:10.1016/j.fertnstert.2010.11.008.PMID21122836.
- ^ "Draft Screening Assessment for The Challenge Phenol, 4,4′-(1-methylethylidene)bis- (Bisphenol A) Chemical Abstracts Service Registry Number 80-05-7".Health Canada.2008. Archived fromthe originalon 5 September 2012.
- ^Ginsberg G, Rice DC (2009)."Does Rapid Metabolism Ensure Negligible Risk from Bisphenol A?".Environmental Health Perspectives.117(11): 1639–1643.doi:10.1289/ehp.0901010.PMC2801165.PMID20049111.
- ^Beronius A, Rudén C, Håkansson H, Hanberg A (April 2010). "Risk to all or none? A comparative analysis of controversies in the health risk assessment of Bisphenol A".Reproductive Toxicology.29(2): 132–46.doi:10.1016/j.reprotox.2009.11.007.PMID19931376.
- ^Woodruff TJ, Zota AR, Schwartz JM (June 2011)."Environmental chemicals in pregnant women in the United States: NHANES 2003–2004".Environmental Health Perspectives.119(6): 878–85.doi:10.1289/ehp.1002727.PMC3114826.PMID21233055.
- ^Brown E (11 November 2010)."Jury still out on BPA, World Health Organization says".Los Angeles Times.Retrieved7 February2011.
- ^"Chemical Used in Plastic Bottles Is Safe, F.D.A. Says".The New York Times.16 August 2008.Retrieved14 March2009.
- ^Szabo L (1 November 2008)."Advisers: FDA decision on safety of BPA 'flawed'".USA Today.Retrieved14 March2009.
- ^"Bisphenol A (BPA): Use in Food Contact Application".News & Events.United States Food & Drug Administration. 30 March 2012.Retrieved14 April2012.
- ^"FDA to Ban BPA from Baby Bottles; Plan Falls Short of Needed Protections: Scientists".Common Dreams. 17 July 2012.Retrieved6 April2015.
- ^"BPA Banned From Baby Bottles".The Huffington Post.17 July 2012.
- ^"CLARITY-BPA Program".NIH National Toxicology Program.23 February 2018.Retrieved5 August2019.
- ^Ostroff S (23 February 2018)."Statement from Stephen Ostroff M.D., Deputy Commissioner for Foods and Veterinary Medicine, on National Toxicology Program draft report on Bisphenol A".U.S. Food and Drug Administration.Retrieved5 August2019.
- ^Vandenberg LN, Ehrlich S, Belcher SM, Ben-Jonathan N, Dolinoy DC, Hugo ER, et al. (October 2013)."Low dose effects of bisphenol A: An integrated review of in vitro, laboratory animal, and epidemiology studies".Endocrine Disruptors.1(1): e26490.doi:10.4161/endo.26490.S2CID82372971.
- ^Rochester JR, Bolden AL (July 2015)."Bisphenol S and F: A Systematic Review and Comparison of the Hormonal Activity of Bisphenol A Substitutes".Environmental Health Perspectives.123(7): 643–50.doi:10.1289/ehp.1408989.PMC4492270.PMID25775505.
- ^Eladak S, Grisin T, Moison D, Guerquin MJ, N'Tumba-Byn T, Pozzi-Gaudin S, et al. (January 2015)."A new chapter in the bisphenol A story: bisphenol S and bisphenol F are not safe alternatives to this compound".Fertility and Sterility.103(1): 11–21.doi:10.1016/j.fertnstert.2014.11.005.PMID25475787.
- ^Tanner EM, Hallerbäck MU, Wikström S, Lindh C, Kiviranta H, Gennings C, et al. (January 2020)."Early prenatal exposure to suspected endocrine disruptor mixtures is associated with lower IQ at age seven".Environment International.134:105185.Bibcode:2020EnInt.13405185T.doi:10.1016/j.envint.2019.105185.PMID31668669.
- ^abDavis KS (1971)."The deadly dust: the unhappy history of DDT".American Heritage Magazine.22(2). Archived fromthe originalon 12 September 2008.Retrieved15 February2009.
- ^"Stockholm Convention on Persistent Organic Pollutants".
- ^Lundholm CD (October 1997). "DDE-induced eggshell thinning in birds: effects of p, p′-DDE on the calcium and prostaglandin metabolism of the eggshell gland".Comp. Biochem. Physiol. C.118(2): 113–28.doi:10.1016/S0742-8413(97)00105-9.PMID9490182.
- ^Szlinder-Richert J, Barska I, Mazerski J, Usydus Z (May 2008). "Organochlorine pesticides in fish from the southern Baltic Sea: levels, bioaccumulation features and temporal trends during the 1995–2006 period".Mar. Pollut. Bull.56(5): 927–40.Bibcode:2008MarPB..56..927S.doi:10.1016/j.marpolbul.2008.01.029.PMID18407298.
- ^Peterle TJ (November 1969)."DDT in Antarctic snow".Nature.224(5219): 620.Bibcode:1969Natur.224..620P.doi:10.1038/224620a0.PMID5346606.S2CID4188794.
- ^Daly GL, Wania F (January 2005). "Organic contaminants in mountains".Environ. Sci. Technol.39(2): 385–98.Bibcode:2005EnST...39..385D.doi:10.1021/es048859u.PMID15707037.S2CID19072832.
- ^Tauber OE, Hughes AB (November 1950). "Effect of DDT ingestion on total cholesterol content of ovaries of white rat".Proc. Soc. Exp. Biol. Med.75(2): 420–2.doi:10.3181/00379727-75-18217.PMID14808278.S2CID252206.
- ^Stoner HB (December 1953). "Effect of 2,2-bis (parachlorophenyl)-1,1-dichloroethane (DDD) on the adrenal cortex of the rat".Nature.172(4388): 1044–5.Bibcode:1953Natur.172.1044S.doi:10.1038/1721044a0.PMID13111250.S2CID4200580.
- ^Tiemann U (April 2008). "In vivo and in vitro effects of the organochlorine pesticides DDT, TCPM, methoxychlor, and lindane on the female reproductive tract of mammals: a review".Reprod. Toxicol.25(3): 316–26.Bibcode:2008RepTx..25..316T.doi:10.1016/j.reprotox.2008.03.002.PMID18434086.
- ^Hallegue D, Rhouma KB, Tébourbi O, Sakly M (April 2003)."Impairment of Testicular Endocrine and Exocrine Functions after Dieldrin Exposure in Adult Rats"(PDF).Polish Journal of Environmental Studies.12(5): 557–562.
- ^abVerhulst SL, Nelen V, Hond ED, Koppen G, Beunckens C, Vael C, et al. (January 2009)."Intrauterine exposure to environmental pollutants and body mass index during the first 3 years of life".Environmental Health Perspectives.117(1): 122–6.doi:10.1289/ehp.0800003.PMC2627855.PMID19165398.
- ^Francis E (1 September 1994)."March/April 2001 Sierra Magazine – Sierra Club".Sierra Magazine.Archived fromthe originalon 20 June 2009.Retrieved14 March2009.
- ^ab"Fox River History of PCBs".Fox River Watch.Clean Water Action Council. Archived fromthe originalon 21 February 2002.Retrieved14 March2009.
- ^Jensen S, Johnels AG, Olsson M, Otterlind G (October 1969). "DDT and PCB in marine animals from Swedish waters".Nature.224(5216): 247–50.Bibcode:1969Natur.224..247J.doi:10.1038/224247a0.PMID5388040.S2CID4182319.
- ^Tang NJ, Liu J, Coenraads PJ, Dong L, Zhao LJ, Ma SW, et al. (April 2008)."Expression of AhR, CYP1A1, GSTA1, c-fos and TGF-alpha in skin lesions from dioxin-exposed humans with chloracne"(PDF).Toxicol. Lett.177(3): 182–7.doi:10.1016/j.toxlet.2008.01.011.hdl:11370/f27e334f-9133-421b-9d78-ff322686e1ae.PMID18329192.S2CID9665736.
- ^abLoomis D, Browning SR, Schenck AP, Gregory E, Savitz DA (October 1997)."Cancer mortality among electric utility workers exposed to polychlorinated biphenyls".Occup Environ Med.54(10): 720–8.doi:10.1136/oem.54.10.720.PMC1128926.PMID9404319.
- ^Brown DP (1987). "Mortality of workers exposed to polychlorinated biphenyls--an update".Arch. Environ. Health.42(6): 333–9.doi:10.1080/00039896.1987.9934355.PMID3125795.S2CID4615591.
- ^Sinks T, Steele G, Smith AB, Watkins K, Shults RA (August 1992). "Mortality among workers exposed to polychlorinated biphenyls".Am. J. Epidemiol.136(4): 389–98.doi:10.1093/oxfordjournals.aje.a116511.PMID1415158.
- ^Grunwald M (1 January 2002)."Monsanto Hid Decades Of Pollution".The Washington Post.Archived fromthe originalon 21 August 2010.Retrieved14 March2009.
- ^Polychlorinated biphenyls and terphenyls,Environmental Health Criteria monograph No. 002, Geneva: World Health Organization, 1976,ISBN92-4-154062-1
- ^abcKodavanti PR (2006)."Neurotoxicity of Persistent Organic Pollutants: Possible Mode(s) of Action and Further Considerations".Dose-Response.3(3): 273–305.doi:10.2203/dose-response.003.03.002.PMC2475949.PMID18648619.
- ^Uemura H, Arisawa K, Hiyoshi M, Satoh H, Sumiyoshi Y, Morinaga K, et al. (September 2008). "Associations of environmental exposure to dioxins with prevalent diabetes among general inhabitants in Japan".Environ. Res.108(1): 63–8.Bibcode:2008ER....108...63U.doi:10.1016/j.envres.2008.06.002.PMID18649880.
- ^Mullerova D, Kopecky J, Matejkova D, Muller L, Rosmus J, Racek J, et al. (December 2008)."Negative association between plasma levels of adiponectin and polychlorinated biphenyl 153 in obese women under non-energy-restrictive regime".Int J Obes (Lond).32(12): 1875–8.doi:10.1038/ijo.2008.169.PMID18825156.
- ^"Effects of human exposure to hormone-disrupting chemicals examined in landmark United Nations report".Science Daily.19 February 2013.Retrieved6 April2015.
- ^Eriksson P, Fischer C, Fredriksson A (December 2006)."Polybrominated diphenyl ethers, a group of brominated flame retardants, can interact with polychlorinated biphenyls in enhancing developmental neurobehavioral defects".Toxicol. Sci.94(2): 302–9.doi:10.1093/toxsci/kfl109.PMID16980691.
- ^abc"The history of plastic".Plastics Division.American Chemistry Council. Archived fromthe originalon 31 December 2008.Retrieved14 March2009.
- ^"Cotton Products Research: Durable Press and Flame Retardant Cotton".National Historic Chemical Landmarks.American Chemical Society.Retrieved21 February2014.
- ^Epidemiology and Statistics Unit (July 2011)."Trends in Tobacco Use"(PDF).American Lung Association.Retrieved2 April2015.
- ^Karter MJ (1 August 2008)."Fire Loss In The United States 2007"(PDF).National Fire Protection Association. Archived fromthe original(PDF)on 7 December 2008.Retrieved14 March2009.
- ^America Burning(PDF)(Report).U.S. Fire Administration.4 May 1973.Retrieved14 March2009.
- ^abcFlame retardants: a general introduction,Environmental Health Criteria monograph No. 192, Geneva: World Health Organization, 1997,ISBN92-4-157192-6
- ^"Great Lakes Chemical Corporation – Company History".Retrieved14 March2009.
- ^"Toxicological review of decabromodiphenyl ether (BDE-209)"(PDF).U.S. Environmental Protection Agency. June 2008.Retrieved14 March2009.
- ^"toxicological review of 2,2′,4,4′-tetrabromodiphenyl ether (BDE-47)"(PDF).U.S. Environmental Protection Agency. 1 June 2008.Retrieved14 March2009.
- ^Betts KS (May 2008)."New thinking on flame retardants".Environmental Health Perspectives.116(5): A210–3.doi:10.1289/ehp.116-a210.PMC2367656.PMID18470294.
- ^Costa LG, Giordano G (November 2007)."Developmental neurotoxicity of polybrominated diphenyl ether (PBDE) flame retardants".Neurotoxicology.28(6): 1047–67.Bibcode:2007NeuTx..28.1047C.doi:10.1016/j.neuro.2007.08.007.PMC2118052.PMID17904639.
- ^Lema SC, Dickey JT, Schultz IR, Swanson P (December 2008)."Dietary exposure to 2,2′,4,4′-tetrabromodiphenyl ether (PBDE-47) alters thyroid status and thyroid hormone-regulated gene transcription in the pituitary and brain".Environmental Health Perspectives.116(12): 1694–9.doi:10.1289/ehp.11570.PMC2599765.PMID19079722.
- ^abFisher JS (March 2004)."Environmental anti-androgens and male reproductive health: focus on phthalates and testicular dysgenesis syndrome".Reproduction.127(3): 305–15.doi:10.1530/rep.1.00025.PMID15016950.
- ^Barrett JR (2005)."Phthalates and Baby Boys: Potential Disruption of Human Genital Development".Environmental Health Perspectives.113(8): A542.doi:10.1289/ehp.113-a542a.JSTOR3436340.PMC1280383.
- ^Kaiser J (October 2005). "Toxicology. Panel finds no proof that phthalates harm infant reproductive systems".Science.310(5747): 422.doi:10.1126/science.310.5747.422a.PMID16239449.S2CID39080713.
- ^"California OKs phthalates ban on children's products".Reuters.15 October 2007.Retrieved14 March2009.
- ^Hileman B (17 October 2007)."California Bans Phthalates In Toys For Children".Chemical & Engineering News.Retrieved14 March2009.
- ^"Children's Safe Product Act – RCW 70.240.020 Lead, Cadmium, and Phthalates"(PDF).Washington State Department of Ecology.12 February 2016. Archived fromthe original(PDF)on 10 February 2017.Retrieved27 April2022.
- ^Feigal DW (12 July 2002)."PVC Devices Containing the Plasticizer DEHP".US FDA/CDRH: FDA Public Health Notification.Food and Drug Administration.Retrieved14 March2009.
- ^Swan SH, Liu F, Hines M, Kruse RL, Wang C, Redmon JB, et al. (November 2009)."Prenatal phthalate exposure and reduced masculine play in boys".Int. J. Androl.33(2): 259–69.doi:10.1111/j.1365-2605.2009.01019.x.PMC2874619.PMID19919614.
- ^Williams MJ, Wiemerslage L, Gohel P, Kheder S, Kothegala LV, Schiöth HB (2016)."Dibutyl Phthalate Exposure Disrupts Evolutionarily Conserved Insulin and Glucagon-Like Signaling in Drosophila Males".Endocrinology.157(6): 2309–21.doi:10.1210/en.2015-2006.PMID27100621.
- ^Steenland K, Fletcher T, Savitz DA (August 2010)."Epidemiologic evidence on the health effects of perfluorooctanoic acid (PFOA)".Environmental Health Perspectives.118(8): 1100–1108.doi:10.1289/ehp.0901827.PMC2920088.PMID20423814.
- ^abcdChaparro-Ortega A, Betancourt M, Rosas P, Vázquez-Cuevas FG, Chavira R, Bonilla E, et al. (February 2018). "Endocrine disruptor effect of perfluorooctane sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA) on porcine ovarian cell steroidogenesis".Toxicology in Vitro.46:86–93.Bibcode:2018ToxVi..46...86C.doi:10.1016/j.tiv.2017.09.030.PMID28982594.
- ^Ben Rhouma B, Kallabi F, Mahfoudh N, Ben Mahmoud A, Engeli RT, Kamoun H, et al. (January 2017). "Novel cases of Tunisian patients with mutations in the gene encoding 17β-hydroxysteroid dehydrogenase type 3 and a founder effect".The Journal of Steroid Biochemistry and Molecular Biology.165(Pt A): 86–94.doi:10.1016/j.jsbmb.2016.03.007.PMID26956191.S2CID25889473.
- ^Tumburu L, Shepard EF, Strand AE, Browdy CL (November 2011). "Effects of endosulfan exposure and Taura Syndrome Virus infection on the survival and molting of the marine penaeid shrimp, Litopenaeus vannamei".Chemosphere.86(9): 912–8.doi:10.1016/j.chemosphere.2011.10.057.PMID22119282.
- ^Burnett ME, Wang SQ (April 2011)."Current sunscreen controversies: a critical review".Photodermatology, Photoimmunology & Photomedicine.27(2): 58–67.doi:10.1111/j.1600-0781.2011.00557.x.PMID21392107.S2CID29173997.
- ^Knobeloch L, Turyk M, Imm P, Schrank C, Anderson H (January 2009). "Temporal changes in PCB and DDE levels among a cohort of frequent and infrequent consumers of Great Lakes sportfish".Environ. Res.109(1): 66–72.Bibcode:2009ER....109...66K.doi:10.1016/j.envres.2008.08.010.PMID18950754.
- ^abNorén K, Meironyté D (2000). "Certain organochlorine and organobromine contaminants in Swedish human milk in perspective of past 20-30 years".Chemosphere.40(9–11): 1111–23.Bibcode:2000Chmsp..40.1111N.doi:10.1016/S0045-6535(99)00360-4.PMID10739053.
- ^Hites RA (February 2004). "Polybrominated diphenyl ethers in the environment and in people: a meta-analysis of concentrations".Environ. Sci. Technol.38(4): 945–56.Bibcode:2004EnST...38..945H.doi:10.1021/es035082g.PMID14998004.S2CID32909270.
- ^Betts KS (February 2002)."Rapidly rising PBDE levels in North America".Environ. Sci. Technol.36(3): 50A–52A.Bibcode:2002EnST...36...50B.doi:10.1021/es022197w.PMID11871568.
- ^Protsiv M, Ley C, Lankester J, Hastie T,Parsonnet J(January 2020)."Decreasing human body temperature in the United States since the industrial revolution".eLife.9.doi:10.7554/eLife.49555.PMC6946399.PMID31908267.
- ^Vancamp P, Demeneix BA (22 July 2020)."Is the Observed Decrease in Body Temperature During Industrialization Due to Thyroid Hormone-Dependent Thermoregulation Disruption?".Frontiers in Endocrinology.11:470.doi:10.3389/fendo.2020.00470.PMC7387406.PMID32793119.
- ^Patisaul HB, Fenton SE, Aylor D (June 2018)."Animal models of endocrine disruption".Best Practice & Research. Clinical Endocrinology & Metabolism.32(3): 283–297.doi:10.1016/j.beem.2018.03.011.PMC6029710.PMID29779582.
- ^Aylor DL, Valdar W, Foulds-Mathes W, Buus RJ, Verdugo RA, Baric RS, et al. (August 2011)."Genetic analysis of complex traits in the emerging Collaborative Cross".Genome Research.21(8): 1213–22.doi:10.1101/gr.111310.110.PMC3149489.PMID21406540.
- ^Threadgill DW, Churchill GA (February 2012)."Ten years of the Collaborative Cross".Genetics.190(2): 291–4.doi:10.1534/genetics.111.138032.PMC3276648.PMID22345604.
- ^Threadgill DW, Hunter KW, Williams RW (April 2002). "Genetic dissection of complex and quantitative traits: from fantasy to reality via a community effort".Mammalian Genome.13(4): 175–8.doi:10.1007/s00335-001-4001-Y.PMID11956758.S2CID17568717.
- ^La Merrill M, Kuruvilla BS, Pomp D, Birnbaum LS, Threadgill DW (September 2009)."Dietary fat alters body composition, mammary development, and cytochrome p450 induction after maternal TCDD exposure in DBA/2J mice with low-responsive aryl hydrocarbon receptors".Environmental Health Perspectives.117(9): 1414–9.doi:10.1289/ehp.0800530.PMC2737019.PMID19750107.
- ^Rocha-Martins M, Cavalheiro GR, Matos-Rodrigues GE, Martins RA (August 2015)."From Gene Targeting to Genome Editing: Transgenic animals applications and beyond".Anais da Academia Brasileira de Ciências.87(2 Suppl): 1323–48.doi:10.1590/0001-3765201520140710.PMID26397828.
- ^Dubois SL, Acosta-Martínez M, DeJoseph MR, Wolfe A, Radovick S, Boehm U, et al. (March 2015)."Positive, but not negative feedback actions of estradiol in adult female mice require estrogen receptor α in kisspeptin neurons".Endocrinology.156(3): 1111–20.doi:10.1210/en.2014-1851.PMC4330313.PMID25545386.
- ^abMcDevitt MA, Glidewell-Kenney C, Jimenez MA, Ahearn PC, Weiss J, Jameson JL, et al. (August 2008)."New insights into the classical and non-classical actions of estrogen: evidence from estrogen receptor knock-out and knock-in mice".Molecular and Cellular Endocrinology.290(1–2): 24–30.doi:10.1016/j.mce.2008.04.003.PMC2562461.PMID18534740.
- ^abStefkovich ML, Arao Y, Hamilton KJ, Korach KS (May 2018)."Experimental models for evaluating non-genomic estrogen signaling".Steroids.133:34–37.doi:10.1016/j.steroids.2017.11.001.PMC5864539.PMID29122548.
- ^Chambliss KL, Wu Q, Oltmann S, Konaniah ES, Umetani M, Korach KS, et al. (July 2010)."Non-nuclear estrogen receptor alpha signaling promotes cardiovascular protection but not uterine or breast cancer growth in mice".The Journal of Clinical Investigation.120(7): 2319–30.doi:10.1172/JCI38291.PMC2898582.PMID20577047.
- ^Li Y, Hamilton KJ, Lai AY, Burns KA, Li L, Wade PA, et al. (March 2014)."Diethylstilbestrol (DES)-stimulated hormonal toxicity is mediated by ERα alteration of target gene methylation patterns and epigenetic modifiers (DNMT3A, MBD2, and HDAC2) in the mouse seminal vesicle".Environmental Health Perspectives.122(3): 262–8.doi:10.1289/ehp.1307351.PMC3948038.PMID24316720.
- ^abcdMcGraw LA, Young LJ (February 2010)."The prairie vole: an emerging model organism for understanding the social brain".Trends in Neurosciences.33(2): 103–9.doi:10.1016/j.tins.2009.11.006.PMC2822034.PMID20005580.
- ^abMcGraw LA, Davis JK, Lowman JJ, ten Hallers BF, Koriabine M, Young LJ, et al. (January 2010)."Development of genomic resources for the prairie vole (Microtus ochrogaster): construction of a BAC library and vole-mouse comparative cytogenetic map".BMC Genomics.11:70.doi:10.1186/1471-2164-11-70.PMC2824727.PMID20109198.
- ^abAdkins-Regan E (2009)."Neuroendocrinology of social behavior".ILAR Journal.50(1): 5–14.doi:10.1093/ilar.50.1.5.PMID19106448.
- ^abAlbers HE (January 2015)."Species, sex and individual differences in the vasotocin/vasopressin system: relationship to neurochemical signaling in the social behavior neural network".Frontiers in Neuroendocrinology.36:49–71.doi:10.1016/j.yfrne.2014.07.001.PMC4317378.PMID25102443.
- ^abYoung LJ, Lim MM, Gingrich B, Insel TR (September 2001). "Cellular mechanisms of social attachment".Hormones and Behavior.40(2): 133–8.doi:10.1006/hbeh.2001.1691.PMID11534973.S2CID7256393.
- ^abModi ME, Young LJ (March 2012)."The oxytocin system in drug discovery for autism: animal models and novel therapeutic strategies".Hormones and Behavior.61(3): 340–50.doi:10.1016/j.yhbeh.2011.12.010.PMC3483080.PMID22206823.
- ^abSullivan AW, Beach EC, Stetzik LA, Perry A, D'Addezio AS, Cushing BS, et al. (October 2014)."A novel model for neuroendocrine toxicology: neurobehavioral effects of BPA exposure in a prosocial species, the prairie vole (Microtus ochrogaster)".Endocrinology.155(10): 3867–81.doi:10.1210/en.2014-1379.PMC6285157.PMID25051448.
- ^Engell MD, Godwin J, Young LJ, Vandenbergh JG (2006). "Perinatal exposure to endocrine disrupting compounds alters behavior and brain in the female pine vole".Neurotoxicology and Teratology.28(1): 103–10.Bibcode:2006NTxT...28..103E.doi:10.1016/j.ntt.2005.10.002.PMID16307867.
- ^Singewald GM, Rjabokon A, Singewald N, Ebner K (March 2011)."The modulatory role of the lateral septum on neuroendocrine and behavioral stress responses".Neuropsychopharmacology.36(4): 793–804.doi:10.1038/npp.2010.213.PMC3055728.PMID21160468.
- ^Rebuli ME, Gibson P, Rhodes CL, Cushing BS, Patisaul HB (November 2016)."Sex differences in microglial colonization and vulnerabilities to endocrine disruption in the social brain".General and Comparative Endocrinology.238:39–46.doi:10.1016/j.ygcen.2016.04.018.PMC5067172.PMID27102938.
- ^abcdSegner H (March 2009). "Zebrafish (Danio rerio) as a model organism for investigating endocrine disruption".Comparative Biochemistry and Physiology. Toxicology & Pharmacology.149(2): 187–95.doi:10.1016/j.cbpc.2008.10.099.PMID18955160.
- ^abReif DM, Truong L, Mandrell D, Marvel S, Zhang G, Tanguay RL (June 2016)."High-throughput characterization of chemical-associated embryonic behavioral changes predicts teratogenic outcomes".Archives of Toxicology.90(6): 1459–1470.doi:10.1007/s00204-015-1554-1.PMC4701642.PMID26126630.
- ^Blanc M, Antczak P, Cousin X, Grunau C, Scherbak N, Rüegg J, et al. (July 2021)."The insecticide permethrin induces transgenerational behavioral changes linked to transcriptomic and epigenetic alterations in zebrafish (Danio rerio)".The Science of the Total Environment.779:146404.Bibcode:2021ScTEn.77946404B.doi:10.1016/j.scitotenv.2021.146404.PMID33752003.S2CID232323014.
- ^Dooley K, Zon LI (June 2000). "Zebrafish: a model system for the study of human disease".Current Opinion in Genetics & Development.10(3): 252–256.doi:10.1016/s0959-437x(00)00074-5.PMID10826982.
- ^Rennekamp AJ, Peterson RT (February 2015)."15 years of zebrafish chemical screening".Current Opinion in Chemical Biology.24:58–70.doi:10.1016/j.cbpa.2014.10.025.PMC4339096.PMID25461724.
- ^Truong L, Reif DM, St Mary L, Geier MC, Truong HD, Tanguay RL (January 2014)."Multidimensional in vivo hazard assessment using zebrafish".Toxicological Sciences.137(1): 212–233.doi:10.1093/toxsci/kft235.PMC3871932.PMID24136191.
- ^Howe K, Clark MD, Torroja CF, Torrance J, Berthelot C, Muffato M, et al. (April 2013)."The zebrafish reference genome sequence and its relationship to the human genome".Nature.496(7446): 498–503.Bibcode:2013Natur.496..498H.doi:10.1038/nature12111.PMC3703927.PMID23594743.
- ^abDietrich JW (2020).Intelligenzabnahme in der Bevölkerung entwickelter Länder: Der negative "Flynn-Effekt" – die unbekannte Seite endokriner Disruptoren. In: Schatz H, Weber M: Endokrinologie – Diabetologie – Stoffwechsel: Neues über Hormone und Metabolismus im Jahre 2019.Hildesheim: Wecom. pp. 35–39.ISBN978-3-00-065109-0.
- ^abDemeneix B, Slama R."Endocrine Disruptors: from Scientific Evidence to Human Health Protection"(PDF).European Parliament.Retrieved6 May2020.
- ^abStacy SL, Papandonatos GD, Calafat AM, Chen A, Yolton K, Lanphear BP, et al. (October 2017)."Early life bisphenol A exposure and neurobehavior at 8years of age: Identifying windows of heightened vulnerability".Environment International.107:258–265.Bibcode:2017EnInt.107..258S.doi:10.1016/j.envint.2017.07.021.PMC5567845.PMID28764921.
- ^Nakiwala D, Peyre H, Heude B, Bernard JY, Béranger R, Slama R, et al. (February 2018)."In-utero exposure to phenols and phthalates and the intelligence quotient of boys at 5 years".Environmental Health.17(1): 17.Bibcode:2018EnvHe..17...17N.doi:10.1186/s12940-018-0359-0.PMC5819230.PMID29458359.
- ^abcdeWayland S, Fenner-Crisp P (March 2016)."Reducing Pesticide Risks: A Half Century of Progress"(PDF).EPA Alumni Association.
- ^Ambrose SG (27 May 2007)."Scientists criticize EPA chemical screening program".Dallas Morning News.Retrieved14 March2009.
- ^Harding AK, Daston GP, Boyd GR, Lucier GW, Safe SH, Stewart J, et al. (August 2006)."Endocrine disrupting chemicals research program of the U.S. Environmental Protection Agency: summary of a peer-review report".Environmental Health Perspectives.114(8): 1276–82.doi:10.1289/ehp.8875.PMC1552001.PMID16882539.
- ^Brussels AN (22 May 2015)."EU dropped pesticide laws due to US pressure over TTIP, documents reveal".Guardian.Retrieved22 May2015.
- ^ab"Sweden to sue EU for delay on hormone disrupting chemicals".22 May 2014.Retrieved10 October2015.
- ^Ing-Marie Olsson (24 November 2014)."The Cost of Inaction: A Socioeconomic analysis of costs linked to effects of endocrine disrupting substances on male reproductive health".Retrieved10 October2015.
- ^"California Biomonitoring Program".State of California. Archived fromthe originalon 16 March 2009.Retrieved14 March2009.
- ^"Fish and Shellfish Advisories and Safe Eating Guidelines".U.S. Environmental Protection Agency.10 November 2014.Retrieved4 March2023.
- ^Guo W, Pan B, Sakkiah S, Yavas G, Ge W, Zou W, et al. (November 2019)."Persistent Organic Pollutants in Food: Contamination Sources, Health Effects and Detection Methods".International Journal of Environmental Research and Public Health.16(22): 4361.doi:10.3390/ijerph16224361.PMC6888492.PMID31717330.
- ^"Green Remediation".United States Environmental Protection Agency. Archived fromthe originalon 15 March 2009.Retrieved14 March2009.
- ^Field JA, Sierra-Alvarez R (September 2008). "Microbial transformation and degradation of polychlorinated biphenyls".Environ. Pollut.155(1): 1–12.Bibcode:2008EPoll.155....1F.doi:10.1016/j.envpol.2007.10.016.PMID18035460.
- ^"Profiles of Green Strategies: Rhizome Collective Inc. Brownfield Site, Austin, TX".Green Remediation.United States Environmental Protection Agency. Archived fromthe originalon 17 March 2009.Retrieved14 March2009.
- ^"Profiles & Case Studies of Green Remediation: Paducah Gaseous Diffusion Plant, Paducah, KY".Green Remediation.United States Environmental Protection Agency. Archived fromthe originalon 17 March 2009.Retrieved14 March2009.
- ^"Case Studies of Green Remediation: Re-Solve, Inc., North Dartmouth, MA".Green Remediation.United States Environmental Protection Agency. Archived fromthe originalon 15 March 2009.Retrieved14 March2009.
- ^Genuis SJ, Beesoon S, Birkholz D, Lobo RA (2012)."Human excretion of bisphenol A: blood, urine, and sweat (BUS) study".J Environ Public Health.2012:1–10.doi:10.1155/2012/185731.PMC3255175.PMID22253637.
- ^Genuis SJ, Beesoon S, Birkholz D (2013)."Biomonitoring and Elimination of Perfluorinated Compounds and Polychlorinated Biphenyls through Perspiration: Blood, Urine, and Sweat Study".ISRN Toxicol.2013:1–7.doi:10.1155/2013/483832.PMC3776372.PMID24083032.
- ^Genuis SJ, Beesoon S, Lobo RA, Birkholz D (2012)."Human elimination of phthalate compounds: blood, urine, and sweat (BUS) study".ScientificWorldJournal.2012:1–10.doi:10.1100/2012/615068.PMC3504417.PMID23213291.
- ^Ohore OE, Zhang S (1 September 2019)."Endocrine disrupting effects of bisphenol A exposure and recent advances on its removal by water treatment systems. A review".Scientific African.5:e00135.Bibcode:2019SciAf...500135O.doi:10.1016/j.sciaf.2019.e00135.ISSN2468-2276.S2CID202079156.
- ^Trasande L, Zoeller RT, Hass U, Kortenkamp A,Grandjean P,Myers JP, et al. (April 2015)."Estimating burden and disease costs of exposure to endocrine-disrupting chemicals in the European union".The Journal of Clinical Endocrinology and Metabolism.100(4): 1245–55.doi:10.1210/jc.2014-4324.PMC4399291.PMID25742516.
- ^Rijk I, Van Duursen M, van den Berg M (2016).Health cost that may be associated with Endocrine Disrupting Chemical – An inventory, evaluation and way forward to assess the potential socio-economic impact of EDC-associated health effects in the EU(PDF)(Report).Universiteit Utrecht,Institute for Risk Assessment Sciences (IRAS).
Further reading
[edit]- Krimsky S (2000).Hormonal chaos: the scientific and social origins of the environmental endocrine hypothesis.Baltimore: Johns Hopkins University Press.ISBN978-0-8018-6279-3.
- Bergman Å, Heindel JJ, Jobling S, Kidd KA, Zoeller RT, eds. (2013).State of the Science of Endocrine Disrupting Chemicals - 2012(PDF).United Nations Environment Programme and the World Health Organization.ISBN978-92-807-3274-0.Archived fromthe original(PDF)on 22 May 2013.Retrieved24 February2013.andISBN978-92-4-150503-1
- Colborn T, Dumanoski D, Meyers P (1996).Our stolen future: are we threatening our fertility, intelligence, and survival?: a scientific detective story.New York: Dutton.ISBN978-0-525-93982-5.
- Andersson N, Arena M, Auteri D, Barmaz S, Grignard E, Kienzler A, et al. (European Chemical Agency (ECHA) and European Food Safety Authority (EFSA) with the technical support of the Joint Research Centre (JRC)) (June 2018)."Guidance for the identification of endocrine disruptors in the context of Regulations (EU) No 528/2012 and (EC) No 1107/2009".EFSA Journal.16(6): e05311.doi:10.2903/j.efsa.2018.5311.PMC7009395.PMID32625944.
- "Revised Guidance Document 150 on Standardised Test Guidelines for Evaluating Chemicals for Endocrine Disruption".OECD.OECD Series on Testing and Assessment. 2018.doi:10.1787/9789264304741-en.ISBN978-92-64-30474-1.S2CID240274054.
- Endocrine Disrupting Chemicals in Freshwater: Monitoring and Regulating Water Quality.OECD Studies on Water. OECD. 12 October 2023.doi:10.1787/5696d960-en.ISBN978-92-64-53751-4.S2CID264081548.Policy Highlights.
External links
[edit]- Endocrine Disruptors,European Commission,2023
- Endocrine Disruption Screening Program,USEnvironmental Protection Agency,April 2023
- Endocrine Disruptor Lists,provided by European authorities, 2023
