Immunoglobulin E
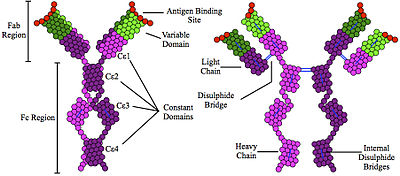


Immunoglobulin E(IgE) is a type ofantibody(orimmunoglobulin(Ig) "isotype") that has been found only inmammals.IgE is synthesised byplasma cells.Monomers of IgE consist of two heavy chains (ε chain) and two light chains, with the ε chain containing four Ig-like constant domains (Cε1–Cε4).[1]IgE is thought to be an important part of theimmune responseagainst infection by certainparasitic worms,includingSchistosoma mansoni,Trichinella spiralis,[2][3]andFasciola hepatica.[4]IgE is also utilized during immune defense against certainprotozoanparasitessuch asPlasmodium falciparum.[5]IgE may have evolved as a defense to protect against venoms.[6][7][8]
IgE also has an essential role intype I hypersensitivity,[9]which manifests in various allergic diseases, such as allergicasthma,most types ofsinusitis,allergic rhinitis,food allergies, and specific types ofchronic urticariaandatopic dermatitis.IgE also plays a pivotal role in responses to allergens, such as:anaphylacticreactions to drugs, bee stings, and antigen preparations used in desensitizationimmunotherapy.
IgE is typically the least abundant isotype:blood serumIgE levels in a non-atopicindividual are only 0.05% of the Ig concentration,[10]compared to 75% for theIgGsat 10 mg/ml. Despite this, it is capable of triggeringanaphylaxis,one of the most rapid and severe immunological reactions.[11]
Discovery
[edit]IgE was simultaneously discovered in 1966 and 1967 by two independent groups:[12]byKimishige Ishizakaand his wifeTeruko Ishizakaat the Children's Asthma Research Institute and Hospital inDenver,Colorado,[13]and byGunnar JohanssonandHans BennichinUppsala,Sweden.[14]Their joint paper was published in April 1969.[15]
Receptors
[edit]IgE primes the IgE-mediated allergic response by binding toFc receptorsfound on the surface ofmast cellsandbasophils.Fc receptors are also found oneosinophils,monocytes,macrophagesandplateletsin humans. There are two types of Fcε receptors:[citation needed]
- FcεRI(type I Fcε receptor), the high-affinity IgE receptor
- FcεRII(type II Fcε receptor), also known as CD23, the low-affinity IgE receptor
IgE can upregulate the expression of both types of Fcε receptors. FcεRI is expressed on mast cells, basophils, and the antigen-presentingdendritic cellsin both mice and humans. Binding ofantigensto IgE already bound by the FcεRI on mast cells causes cross-linking of the bound IgE and the aggregation of the underlying FcεRI, leading todegranulation(the release of mediators) and the secretion of several types of type 2cytokineslikeinterleukin(IL)-3andstem cell factor(SCF), which both help the mast cells survive and accumulate in tissue, andIL-4,IL-5,IL-13,andIL-33,which in turn activate group 2-innate lymphoid cells (ILC2or natural helper cells). Basophils share a common haemopoietic progenitor with mast cells; upon the cross-linking of their surface bound IgE by antigens, also release type 2 cytokines, including IL-4 and IL-13, and other inflammatory mediators. The low-affinity receptor (FcεRII) is always expressed onB cells;but IL-4 can induce its expression on the surfaces of macrophages, eosinophils, platelets, and someT cells.[16][17]
Function
[edit]Parasite hypothesis
[edit]The IgE isotype has co-evolved with basophils and mast cells in the defence against parasites like helminths (like Schistosoma) but may be also effective in bacterial infections.[18] Epidemiological research shows that IgE level is increased when infected bySchistosoma mansoni,[19]Necator americanus,[20]andnematodes[21]in humans. It is most likely beneficial in removal of hookworms from the lung.[citation needed]
Toxin hypothesis of allergic disease
[edit]In 1981Margie Profetsuggested that allergic reactions have evolved as a last line of defense to protect againstvenoms.[6]Although controversial at the time, new work supports some of Profet’s thoughts on the adaptive role of allergies as a defense against noxious toxins.[7]
In 2013 it emerged that IgE-antibodies play an essential role in acquired resistance tohoney bee[8]andRussell's vipervenoms.[8][22]The authors concluded that "a small dose of bee venom conferred immunity to a much larger, fatal dose" and "this kind of venom-specific, IgE-associated, adaptive immune response developed, at least in evolutionary terms, to protect the host against potentially toxic amounts of venom, such as would happen if the animal encountered a whole nest of bees, or in the event of a snakebite".[8][23][24]The major allergen of bee venom (phospholipase A2) induces aTh2immune responses, associated with production of IgE antibodies, which may "increase the resistance of mice to challenge with potentially lethal doses".[25]
Cancer
[edit]Although it is not yet well understood, IgE may play an important role in the immune system's recognition ofcancer,[26]in which the stimulation of a strong cytotoxic response against cells displaying only small amounts of early cancer markers would be beneficial. If this were the case, anti-IgE treatments such asomalizumab(for allergies) might have some undesirable side effects. However, a recent study, which was performed based on pooled analysis using comprehensive data from 67 phase I to IV clinical trials of omalizumab in various indications, concluded that a causal relationship between omalizumab therapy and malignancy is unlikely.[27]
Role in disease
[edit]Atopicindividuals can have up to ten times the normal level of IgE in their blood (as do sufferers ofhyper-IgE syndrome). However, this may not be a requirement for symptoms to occur as has been seen in asthmatics with normal IgE levels in their blood—recent research has shown that IgE production can occur locally in the nasal mucosa.[28]
IgE that can specifically recognise anallergen(typically this is a protein, such asdust miteDer p 1,catFel d 1,grass orragweedpollen, food protein, etc.) has a unique long-lived interaction with its high-affinityreceptorFcεRI so thatbasophilsandmast cells,capable of mediating inflammatory reactions, become "primed", ready to release chemicals likehistamine,leukotrienes,and certain interleukins. These chemicals cause many of the symptoms we associate with allergy, such as airway constriction inasthma,local inflammation ineczema,increasedmucussecretion inallergic rhinitis,and increased vascular permeability, it is presumed, to allow other immune cells to gain access to tissues, but which can lead to a potentially fatal drop in blood pressure as inanaphylaxis.[citation needed]
IgE is known to be elevated in various autoimmune disorders such asSLE,rheumatoid arthritis(RA), andpsoriasis,and is theorized to be of pathogenetic importance in SLE and RA by eliciting a hypersensitivity reaction.[29][30]
Regulation of IgE levels through control of B cell differentiation to antibody-secretingplasma cellsis thought to involve the "low-affinity" receptor FcεRII, orCD23.[31]CD23may also allow facilitated antigen presentation, an IgE-dependent mechanism wherebyB cellsexpressingCD23are able to present allergen to (and stimulate) specificT helper cells,causing the perpetuation of a Th2 response, one of the hallmarks of which is the production of more antibodies.[32]
Role in diagnosis
[edit]Diagnosis of allergy is most often done by reviewing a person's medical history and finding a positive result for the presence of allergen specific IgE when conducting a skin or blood test.[33]Specific IgE testing is the proven test for allergy detection; evidence does not show that indiscriminate IgE testing or testing for immunoglobulin G (IgG) can support allergy diagnosis.[34]
Drugs targeting the IgE pathway
[edit]Currently, allergic diseases and asthma are usually treated with one or more of the following drugs: (1)antihistaminesandantileukotrienes,which antagonize the inflammatory mediators histamine and leukotrienes, (2) local or systemic (oral or injectable)corticosteroids,which suppress a broad spectrum of inflammatory mechanisms, (3) short or long-actingbronchodilators,which relax smooth muscle of constricted airway in asthma, or (4)mast cell stabilizers,which inhibit thedegranulationof mast cells that is normally triggered by IgE-binding atFcεRI.Long-term uses of systemic corticosteroids are known to cause many serious side effects and are advisable to avoid, if alternative therapies are available.[citation needed]
IgE, the IgE synthesis pathway, and the IgE-mediated allergic/inflammatory pathway are all important targets in intervening with the pathological processes of allergy, asthma, and other IgE-mediated diseases. The B lymphocyte differentiation and maturation pathway that eventually generate IgE-secreting plasma cells go through the intermediate steps of IgE-expressing B lymphoblasts and involves the interaction with IgE-expressing memory B cells.Tanox,a biotech company based in Houston, Texas, proposed in 1987 that by targeting membrane-bound IgE (mIgE) on B lymphoblast and memory B cells, those cells can be lysed or down-regulated, thus achieving the inhibition of the production of antigen-specific IgE and hence a shift of immune balance toward non-IgE mechanisms.[35]Two approaches targeting the IgE pathway were evolved and both are in active development. In the first approach, theanti-IgEantibody drugomalizumab(trade nameXolair) recognises IgE not bound to its receptors and is used to neutralise or mop-up existing IgE and prevent it from binding to the receptors on mast cells and basophils. Xolair has been approved in many countries for treating severe, persistent allergic asthma. It has also been approved in March 2014 in the European Union[36]and the U. S.[37]for treatingchronic spontaneous urticaria,which cannot be adequately treated with H1-antihistamines.In the second approach, antibodies specific for a domain of 52 amino acid residues, referred to as CεmX or M1’ (M1 prime), present only on human mIgE on B cells and not on free, soluble IgE, have been prepared and are under clinical development for the treatment of allergy and asthma.[38][39]An anti-M1’ humanized antibody,quilizumab,is in phase IIb clinical trial.[40][41]
In 2002, researchers at theRandall Division of Cell and Molecular Biophysicsdetermined the structure of IgE.[42]Understanding of this structure (which is atypical of other isotypes in that it is highly bent and asymmetric) and of the interaction of IgE with receptor FcεRI will enable development of a new generation of allergy drugs that seek to interfere with the IgE-receptor interaction. It may be possible to design treatments cheaper than monoclonal antibodies (for instance, small molecule drugs) that use a similar approach to inhibit binding of IgE to its receptor.[citation needed]
References
[edit]- ^"Antibody structure".Archived fromthe originalon September 6, 2008.
- ^Erb KJ (May 2007)."Helminths, allergic disorders and IgE-mediated immune responses: where do we stand?".European Journal of Immunology.37(5): 1170–3.doi:10.1002/eji.200737314.PMID17447233.S2CID24519249.
- ^Watanabe N, Bruschi F, Korenaga M (April 2005). "IgE: a question of protective immunity in Trichinella spiralis infection".Trends in Parasitology.21(4): 175–8.doi:10.1016/j.pt.2005.02.010.PMID15780839.
- ^Pfister K, Turner K, Currie A, Hall E, Jarrett EE (November 1983). "IgE production in rat fascioliasis".Parasite Immunology.5(6): 587–93.doi:10.1111/j.1365-3024.1983.tb00775.x.PMID6657297.S2CID1530964.
- ^Duarte J, Deshpande P, Guiyedi V, Mécheri S, Fesel C, Cazenave PA, et al. (January 2007)."Total and functional parasite specific IgE responses in Plasmodium falciparum-infected patients exhibiting different clinical status".Malaria Journal.6:1.doi:10.1186/1475-2875-6-1.PMC1781948.PMID17204149.
- ^abProfet M (March 1991). "The function of allergy: immunological defense against toxins".The Quarterly Review of Biology.66(1): 23–62.doi:10.1086/417049.PMID2052671.S2CID5648170.
- ^abPalm NW, Rosenstein RK, Medzhitov R (April 2012)."Allergic host defences".Nature.484(7395): 465–72.Bibcode:2012Natur.484..465P.doi:10.1038/nature11047.PMC3596087.PMID22538607.
- ^abcdMarichal T, Starkl P, Reber LL, Kalesnikoff J, Oettgen HC, Tsai M, et al. (November 2013)."A beneficial role for immunoglobulin E in host defense against honeybee venom".Immunity.39(5): 963–75.doi:10.1016/j.immuni.2013.10.005.PMC4164235.PMID24210352.
- ^Gould HJ, Sutton BJ, Beavil AJ, Beavil RL, McCloskey N, Coker HA, et al. (2003). "The biology of IGE and the basis of allergic disease".Annual Review of Immunology.21:579–628.doi:10.1146/annurev.immunol.21.120601.141103.PMID12500981.
- ^Winter WE, Hardt NS, Fuhrman S (September 2000). "Immunoglobulin E: importance in parasitic infections and hypersensitivity responses".Archives of Pathology & Laboratory Medicine.124(9): 1382–5.doi:10.5858/2000-124-1382-IE.PMID10975945.
- ^Reber, LL; Hernandez, JD; Galli, SJ (August 2017)."The pathophysiology of anaphylaxis".The Journal of Allergy and Clinical Immunology.140(2): 335–348.doi:10.1016/j.jaci.2017.06.003.PMC5657389.PMID28780941.
- ^Stanworth DR (February 1993)."The discovery of IgE".Allergy.48(2): 67–71.doi:10.1111/j.1398-9995.1993.tb00687.x.PMID8457034.S2CID36262710.
- ^Ishizaka K, Ishizaka T, Hornbrook MM (July 1966)."Physico-chemical properties of human reaginic antibody. IV. Presence of a unique immunoglobulin as a carrier of reaginic activity".Journal of Immunology.97(1): 75–85.doi:10.4049/jimmunol.97.1.75.PMID4162440.S2CID255620017.
- ^Johansson SG, Bennich H (October 1967)."Immunological studies of an atypical (myeloma) immunoglobulin".Immunology.13(4): 381–94.PMC1409218.PMID4168094.
- ^Ishizaka T, Ishizaka K, Johansson SG, Bennich H (April 1969)."Histamine release from human leukocytes by anti-gamma E antibodies".Journal of Immunology.102(4): 884–92.doi:10.4049/jimmunol.102.4.884.PMID4181251.S2CID255338552.Retrieved2016-02-29.
- ^Ewart MA, Ozanne BW, Cushley W (May 2002)."The CD23a and CD23b proximal promoters display different sensitivities to exogenous stimuli in B lymphocytes".Genes and Immunity.3(3): 158–64.doi:10.1038/sj.gene.6363848.PMID12070780.
- ^Novak N, Kraft S, Bieber T (December 2001). "IgE receptors".Current Opinion in Immunology.13(6): 721–6.doi:10.1016/s0952-7915(01)00285-0.PMID11677096.
- ^Abraham, Soman N.; St. John, Ashley L. (June 2010)."Mast cell-orchestrated immunity to pathogens".Nature Reviews Immunology.10(6): 440–452.doi:10.1038/nri2782.ISSN1474-1741.PMC4469150.PMID20498670.
- ^Rihet P, Demeure CE, Bourgois A, Prata A, Dessein AJ (November 1991). "Evidence for an association between human resistance to Schistosoma mansoni and high anti-larval IgE levels".European Journal of Immunology.21(11): 2679–86.doi:10.1002/eji.1830211106.PMID1936116.S2CID1103861.
- ^Pritchard DI, Quinnell RJ, Walsh EA (February 1995). "Immunity in humans to Necator americanus: IgE, parasite weight and fecundity".Parasite Immunology.17(2): 71–5.doi:10.1111/j.1365-3024.1995.tb00968.x.PMID7761110.S2CID41222700.
- ^Turner JD, et al. (June 2005)."Allergen-specific IgE and IgG4 are markers of resistance and susceptibility in a human intestinal nematode infection".Microbes and Infection.7(7–8): 990–6.doi:10.1016/j.micinf.2005.03.036.PMID15961339.
- ^Starkl P, Marichal T, Gaudenzio N, Reber LL, Sibilano R, Tsai M, Galli SJ (January 2016)."IgE antibodies, FcεRIα, and IgE-mediated local anaphylaxis can limit snake venom toxicity".The Journal of Allergy and Clinical Immunology.137(1): 246–257.e11.doi:10.1016/j.jaci.2015.08.005.PMC4715494.PMID26410782.
- ^Sharlach, Molly (24 October 2013)."Bee sting allergy could be a defense response gone haywire, scientists say".Stanford Medicine News Center.Retrieved20 November2019.
- ^Foley, James A. (25 October 2013)."Severe Allergies to Bee Stings may be Malfunctioning Evolutionary Response".Nature World News.
- ^Tsai M, Starkl P, Marichal T, Galli SJ (October 2015)."Testing the 'toxin hypothesis of allergy': mast cells, IgE, and innate and acquired immune responses to venoms".Current Opinion in Immunology.36:80–7.doi:10.1016/j.coi.2015.07.001.PMC4593748.PMID26210895.
- ^Karagiannis SN, Wang Q, East N, Burke F, Riffard S, Bracher MG, et al. (April 2003). "Activity of human monocytes in IgE antibody-dependent surveillance and killing of ovarian tumor cells".European Journal of Immunology.33(4): 1030–40.doi:10.1002/eji.200323185.PMID12672069.S2CID29495137.
- ^Busse W, Buhl R, Fernandez Vidaurre C, Blogg M, Zhu J, Eisner MD, Canvin J (April 2012). "Omalizumab and the risk of malignancy: results from a pooled analysis".The Journal of Allergy and Clinical Immunology.129(4): 983–9.e6.doi:10.1016/j.jaci.2012.01.033.PMID22365654.
- ^Takhar P, Smurthwaite L, Coker HA, Fear DJ, Banfield GK, Carr VA, et al. (April 2005)."Allergen drives class switching to IgE in the nasal mucosa in allergic rhinitis".Journal of Immunology.174(8): 5024–32.doi:10.4049/jimmunol.174.8.5024.PMID15814733.
- ^Permin H, Wiik A (October 1978). "The prevalence of IgE antinuclear antibodies in rheumatoid arthritis and systemic lupus erythematosus".Acta Pathologica et Microbiologica Scandinavica, Section C.86C(5): 245–9.doi:10.1111/j.1699-0463.1978.tb02587.x.PMID309705.S2CID13737751.
- ^Elkayam O, Tamir R, Pick AI, Wysenbeek A (January 1995). "Serum IgE concentrations, disease activity, and atopic disorders in systemic lupus erythematosus".Allergy.50(1): 94–6.PMID7741196.
- ^Conrad DH, Ford JW, Sturgill JL, Gibb DR (September 2007). "CD23: an overlooked regulator of allergic disease".Current Allergy and Asthma Reports.7(5): 331–7.doi:10.1007/s11882-007-0050-y.PMID17697638.S2CID24816595.
- ^Holm J, Willumsen N, Würtzen PA, Christensen LH, Lund K (April 2011). "Facilitated antigen presentation and its inhibition by blocking IgG antibodies depends on IgE repertoire complexity".The Journal of Allergy and Clinical Immunology.127(4): 1029–37.doi:10.1016/j.jaci.2011.01.062.PMID21377718.
- ^Cox L, Williams B, Sicherer S, Oppenheimer J, Sher L, Hamilton R, Golden D (December 2008). "Pearls and pitfalls of allergy diagnostic testing: report from the American College of Allergy, Asthma and Immunology/American Academy of Allergy, Asthma and Immunology Specific IgE Test Task Force".Annals of Allergy, Asthma & Immunology.101(6): 580–92.doi:10.1016/S1081-1206(10)60220-7.PMID19119701.
- ^American Academy of Allergy, Asthma, and Immunology."Five Things Physicians and Patients Should Question"(PDF).Choosing Wisely: An Initiative of the ABIM Foundation.Archived fromthe original(PDF)on November 3, 2012.RetrievedAugust 14,2012.
{{cite journal}}:CS1 maint: multiple names: authors list (link) - ^Chang TW, Wu PC, Hsu CL, Hung AF (2007).Anti‐IgE Antibodies for the Treatment of IgE‐Mediated Allergic Diseases.Advances in Immunology. Vol. 93. pp. 63–119.doi:10.1016/S0065-2776(06)93002-8.ISBN9780123737076.PMID17383539.
- ^"Novartis announces Xolair® approved in EU as first and only licensed therapy for chronic spontaneous urticaria (CSU) patients unresponsive to antihistamines".Novartis. 2014-03-06. Archived fromthe originalon 2014-12-18.Retrieved2014-12-04.
- ^"Novartis announces US FDA approval of Xolair® for chronic idiopathic urticaria (CIU)".Novartis. 2014-03-21. Archived fromthe originalon 2014-12-03.Retrieved2014-12-04.
- ^Chen JB, Wu PC, Hung AF, Chu CY, Tsai TF, Yu HM, et al. (February 2010)."Unique epitopes on C epsilon mX in IgE-B cell receptors are potentially applicable for targeting IgE-committed B cells".Journal of Immunology.184(4): 1748–56.doi:10.4049/jimmunol.0902437.PMID20083663.
- ^Brightbill HD, Jeet S, Lin Z, Yan D, Zhou M, Tan M, et al. (June 2010)."Antibodies specific for a segment of human membrane IgE deplete IgE-producing B cells in humanized mice".The Journal of Clinical Investigation.120(6): 2218–29.doi:10.1172/JCI40141.PMC2877936.PMID20458139.
- ^"MEMP1972A".ClinicalTrials.gov.U.S.National Institutes of Health.Retrieved2014-12-04.
- ^Gauvreau GM, Harris JM, Boulet LP, Scheerens H, Fitzgerald JM, Putnam WS, et al. (July 2014). "Targeting membrane-expressed IgE B cell receptor with an antibody to the M1 prime epitope reduces IgE production".Science Translational Medicine.6(243): 243ra85.doi:10.1126/scitranslmed.3008961.PMID24990880.S2CID41593528.
- ^Wan T, Beavil RL, Fabiane SM, Beavil AJ, Sohi MK, Keown M, et al. (July 2002). "The crystal structure of IgE Fc reveals an asymmetrically bent conformation".Nature Immunology.3(7): 681–6.doi:10.1038/ni811.PMID12068291.S2CID32347451.
