Mercury(II) fulminate
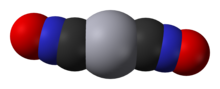
| |
| |

| |
| Names | |
|---|---|
| IUPAC name
Mercury(II) fulminate
| |
| Systematic IUPAC name
Dioxycyanomercury | |
| Other names
Fulminated Mercury
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.010.053 |
PubChemCID
|
|
| UNII | |
CompTox Dashboard(EPA)
|
|
| |
| |
| Properties | |
| C2N2O2Hg | |
| Molar mass | 284.624 g/mol |
| Appearance | Grey, Pale Brown, or White Crystalline solid |
| Density | 4.42 g/cm3 |
| Melting point | 160 °C (320 °F; 433 K) |
| Boiling point | 356.6 °C (673.9 °F; 629.8 K) |
| slightly soluble | |
| Solubility | soluble inethanol,ammonia |
| Explosive data | |
| Shock sensitivity | High |
| Friction sensitivity | High |
| Detonation velocity | 4250 m/s |
| Hazards | |
| Occupational safety and health(OHS/OSH): | |
Main hazards
|
Highly Toxic, Shock Sensitive Explosive |
| NFPA 704(fire diamond) | |
| 170 °C (338 °F; 443 K) | |
Except where otherwise noted, data are given for materials in theirstandard state(at 25 °C [77 °F], 100 kPa).
| |
Mercury(II) fulminate,orHg(CNO)2,is aprimary explosive.It is highly sensitive tofriction,heat andshockand is mainly used as a trigger for other explosives inpercussion capsanddetonators.Mercury(II) cyanate, though its chemical formula is identical, has a different atomic arrangement; thecyanateandfulminateanionsareisomers.
First used as apriming compositionin small copper caps beginning in the 1820s, mercury fulminate quickly replacedflintsas a means to igniteblack powdercharges inmuzzle-loadingfirearms.Later, during the late 19th century and most of the 20th century, mercury fulminate became widely used inprimersforself-contained rifle and pistol ammunition;it was the only practical detonator for firing projectiles until the early 20th century.[1]Mercury fulminate has the distinct advantage overpotassium chlorateof being non-corrosive, but it is known to weaken with time, by decomposing into its constituent elements. The reduced mercury which results forms amalgams with cartridge brass, weakening it, as well. Today, mercury fulminate has been replaced in primers by more efficient chemical substances. These are non-corrosive, less toxic, and more stable over time; they includelead azide,lead styphnate,andtetrazenederivatives. In addition, none of these compounds requiresmercuryfor manufacture, supplies of which can be unreliable in wartime.
Preparation[edit]
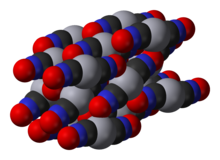
Mercury(II) fulminate is prepared by dissolvingmercuryinnitric acidand addingethanolto the solution. It was first prepared byEdward Charles Howardin 1800.[2][1]Thecrystal structureof this compound was determined only in 2007.[3]
Silver fulminatecan be prepared in a similar way, but thissaltis even more unstable than mercury fulminate; it can explode even under water and is impossible to accumulate in large amounts because it detonates under its own weight.[4]
Decomposition[edit]
The thermal decomposition of mercury(II) fulminate can begin at temperatures as low as 100 °C, though it proceeds at a much higher rate with increasing temperature.[5]
A possible reaction for the decomposition of mercury(II) fulminate yieldscarbon dioxidegas,nitrogengas, and a combination of relatively stable mercury salts.
- 4 Hg(CNO)2→ 2 CO2+ N2+ HgO + 3 Hg(OCN)CN
- Hg(CNO)2→ 2 CO + N2+ Hg
- Hg(CNO)2→:Hg(OCN)2(cyanateor / andisocyanate)
- 2 Hg(CNO)2→ 2 CO2+ N2+ Hg + Hg(CN)2(mercury(II) cyanide)
See also[edit]
References[edit]
- ^abWisniak, Jaime (2012)."Edward Charles Howard. Explosives, meteorites, and sugar".Educación Química.23(2). Universidad Nacional Autonoma de Mexico: 230–239.doi:10.1016/s0187-893x(17)30114-3.ISSN0187-893X.
- ^Edward Howard (1800). "On a New Fulminating Mercury".Philosophical Transactions of the Royal Society of London.90(1): 204–238.doi:10.1098/rstl.1800.0012.S2CID138658702.
- ^W. Beck; J. Evers; M. Göbel; G. Oehlinger; T. M. Klapötke (2007)."The Crystal and Molecular Structure of Mercury Fulminate (Knallquecksilber)".Zeitschrift für anorganische und allgemeine Chemie.633(9): 1417–1422.doi:10.1002/zaac.200700176.
- ^"The Sciences - Fulminating Substances".Scientific American.11 June 1853.
- ^W. E. Garner & H. R. Hailes (1933)."Thermal decomposition and detonation of mercury fulminate".Proceedings of the Royal Society of London.139(1–3): 1–40.Bibcode:1933CP....334..128S.doi:10.1098/rspa.1933.0040.


