Enantiomer
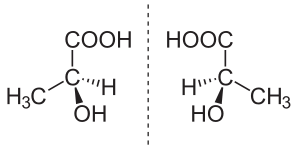
Inchemistry,anenantiomer(/[invalid input: 'icon'][invalid input: 'ɨ']ˈnæntɪ.[invalid input: 'ɵ']mər/ə-NAN-tee-ə-mər;from the Greekἐνάντιος,opposite,andμέρος,part or portion) is one of twostereoisomersthat aremirror imagesof each other that are "non-superposable" (not identical), much as one's left and righthandsare "the same" but opposite[1](It can be clearly understood if you try to place your hands one over the other without touching the back or palm of the left to the same of the right. You observe that the thumb of one is always over the little-finger of the other, thus explaining the non-superimposable or non-coincident property known aschirality)
Organic compounds that contain an asymmetric (chiral) Carbon usually have two non-superimposable structures. These two structures are mirror images of each other and are thus, commonly calledenantiomorphs(enantio= opposite;morph= form) Hence, optical isomerism (which occurs due to this samemirror-imageproperties) is now commonly referred to asenantiomerism
Enantiopure compoundsrefer to samples having,within the limits of detection,molecules of only onechirality.[2]
Enantiomers have, when present in a symmetric environment, identical chemical and physical properties except for their ability to rotateplane-polarized light(+/−) by equal amounts but in opposite directions (although the polarized light can be considered an asymmetric medium). A mixture ofequal partsof an optically active isomer and its enantiomer is termedracemicand has zero net rotation of plane-polarized light.
Enantiomers of each other often show different chemical reactions with other substances that are also enantiomers. Since many molecules in the body of living beings are enantiomers themselves, there is often a marked difference in the effects of two enantiomers on living beings. Indrugs,for example, often only one of a drug's enantiomers is responsible for the desired physiologic effects, while the other enantiomer is less active, inactive, or sometimes even responsible foradverse effects(unwanted side effects).
Owing to this discovery, drugs composed of only one enantiomer ( "entantiopure" ) can be developed to enhance the pharmacological efficacy and sometimes do away with some side effects. An example of this kind of drug iseszopiclone(Lunesta), which is enantiopure and therefore is given in doses that are exactly 1/2 of the older,racemicmixture calledzopiclone.In the case of eszopiclone, the S enantiomer is responsible for all the desired effects, though the other enantiomer seems to be inactive; while an individual must take 2 mg of zopiclone to get the same therapeutic benefit as they would receive from 1 mg of eszopiclone, that appears to be the only difference between the two drugs, and this may be an example of a pharmaceutical company taking advantage of laws that allow them to be issued a brand new patent for the enantiopure version of an existing drug. This leads to a great deal of profit that would otherwise be cut into by generics, as patents on zopiclone expired long ago and it is therefore available in generic form.
Naming conventions
Criterion of Enantiomerism
Most compounds which contain one or more asymmetric Carbon atoms show enantiomerism. But this is not always true.
There are a few known compounds whichdohave asymmetric Carbons but beingnon-dissymetricdo not show enantiomerism. Thusmesotartaric acid hastwoasymmetric Carbons but is still optically inactive. Similarly,trans-cyclohexane - 1,4 - dicarboxylic acid has asymmetric Carbons but also has acentre of symmetry(thereby making it a non-dissymmetric compound) and therefore exhibits no enantiomerism.
Examples


An example of such an enantiomer is thesedativethalidomide.It was sold in a number of countries across the world from 1957 until 1961, when it was withdrawn from the market after being found to be a cause of birth defects.
In theherbicidemecoprop,thecarboxyl groupand the hydrogen atom on the central C-atom are exchanged (with the screen asplane of symmetry). After rotating one of the isomers 180 degrees (in the same plane), the two are still mirror images of each other. The mirror image of each enantiomer is superposable on the other enantiomer.
Another example is the antidepressant drugsescitalopramandcitalopram.Citalopram is aracemate[1:1 mixture of (S)-citalopram and (R)-citalopram]; escitalopram [(S)-citalopram] is a pure enantiomer. The dosages for escitalopram are typically 1/2 of those for citalopram.
Enantioselective preparations
There are two main strategies for the preparation of enantiopure compounds. The first is known as chiral resolution. This method involves preparing the compound inracemicform, and separating it into its isomers. In his pioneering work,Louis Pasteurwas able to isolate the isomers oftartaric acidbecause they crystallize from solution as crystals each with a different symmetry. A less common method is byenantiomer self-disproportionation.
The second strategy is asymmetric synthesis: the use of various techniques to prepare the desired compound in highenantiomeric excess.Techniques encompassed include the use of chiral starting materials (chiral pool synthesis), the use ofchiral auxiliariesandchiral catalysts,and the application ofasymmetric induction.The use of enzymes (biocatalysis) may also produce the desired compound.
Enantioconvergent synthesisis the synthesis of one enantiomer from a racemic precursor molecule utilizing both enantiomers. Thus, the two enantiomers of the reactant produce a single enantiomer of product.
Enantiopure medications
Advances in industrial chemical processes have made it economical for pharmaceutical manufacturers to take drugs that were originally marketed as aracemic mixtureand market the individual enantiomers. In some cases, the enantiomers have genuinely different effects. In other cases, there may be no clinical benefit to the patient. In some jurisdictions, single-enantiomer drugs are separately patentable from the racemic mixture.[3]It is possible that both enantiomers are active. Or, it may be that only one is active, in which case separating the mixture has no objective benefits, but extends the drug's patentability.[4]
Quasi-enantiomers
These are not strictly enantiomers, but they behave as if they are.Quasi-enantiomers have applications in parallel kinetic resolution (seekinetic resolution).[5]
See also
- Enantiopure drug
- Stereochemistry
- Dynamic stereochemistry
- Chirality (chemistry)
- Diastereomers
- Stereogenic
- Atropisomerism
- Antipode (chemistry)
External Links
References
- ^IUPAC,Compendium of Chemical Terminology,2nd ed. (the "Gold Book" ) (1997). Online corrected version: (2006–) "enantiomer".doi:10.1351/goldbook.E02069
- ^IUPAC,Compendium of Chemical Terminology,2nd ed. (the "Gold Book" ) (1997). Online corrected version: (2006–) "enantiomerically pure (enantiopure)".doi:10.1351/goldbook.E02072
- ^http://www.ema.europa.eu/ema/index.jsp?curl=pages/news_and_events/news/2009/11/news_detail_000083.jsp&jsenabled=true
- ^Merrill Goozner (2004).The $800 Million Pill: The Truth Behind the Cost of New Drugs(excerpt).University of California Press.ISBN0-520-23945-8.
- ^G.S. Coumbarides, M. Dingjan, J. Eames, A. Flinn, J. Northen and Y. Yohannes, Tetrahedron Lett. 46 (2005), p. 2897er({{pro
