Sugar acid
Appearance
Inorganic chemistry,asugar acidoracidic sugaris amonosaccharidewith acarboxyl groupat one end or both ends of itschain.[1]
Main classes of sugar acids include:
- Aldonic acids,in which thealdehyde group(−CH=O) located at the initial end (position 1) of analdoseis oxidized.
- Ulosonic acids,in which thehydroxymethyl group(−CH2OH) at the initial end of a 2-ketoseis oxidized creating an α-ketoacid.
- Uronic acids,in which the−CH2OHgroup at the terminal end of an aldose or ketose is oxidized.
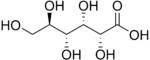
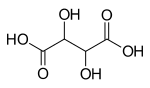
- Aldaric acids,in which both ends (−CH=Oand−CH2OH) of an aldose are oxidized.
 |
 |
 |
Examples
[edit]Examples of sugar acids include:
- Aldonic acids
- Glyceric acid(3C)
- Xylonic acid(5C)
- Gluconic acid(6C)
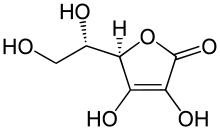
- Ascorbic acid[2](6C, unsaturated lactone)
- Ulosonic acids
- Neuraminic acid(5-amino-3,5-dideoxy-D-glycero-D-galacto-non-2-ulosonic acid)
- Ketodeoxyoctulosonic acid(KDO or 3-deoxy-D-manno-oct-2-ulosonic acid)
- Uronic acids
- Glucuronic acid(6C)
- Galacturonic acid(6C)
- Iduronic acid(6C)
- Aldaric acids
- Tartaric acid(4C)
- meso-Galactaric acid (Mucic acid) (6C)
- D-Glucaric acid (Saccharic acid) (6C)
 |
 |
References
[edit]- ^Robyt, J.F. (1998).Essentials of carbohydrate chemistry.New York: Springer.ISBN0-387-94951-8.
- ^Davies Michael B.; Austin John; Partridge David A. (1991).Vitamin C: Its Chemistry and Biochemistry.The Royal Society of Chemistry. p. 48.ISBN0-85186-333-7.
External links
[edit]- Sugar+Acidsat the U.S. National Library of MedicineMedical Subject Headings(MeSH)

