Trifarotene
 | |
| Clinical data | |
|---|---|
| Trade names | Aklief |
| Other names | CD5789 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a620004 |
| License data | |
| Pregnancy category | |
| Routes of administration | Topical |
| Drug class | Skin and mucous membrane agents |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChemCID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard(EPA) | |
| ECHA InfoCard | 100.278.901 |
| Chemical and physical data | |
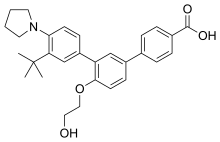
| Formula | C29H33NO4 |
| Molar mass | 459.586g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Trifarotene,sold under the brand nameAklief,is amedicationfor thetopical treatmentofacne vulgaris.[6][8]It is aretinoid;[6][9]specifically, a fourth-generation selectiveretinoic acid receptor (RAR)-γagonist.[10]
Trifarotene was grantedorphan drugdesignation for the treatment of congenitalichthyosisby both the U.S.Food and Drug Administration(FDA) and theEuropean Medicines Agency(EMA).[11][12]It was approved for medical use in the United States in October 2019.[8][13][14]In December 2019, its labelling and package leaflet text received a decentralised approval for 16 European countries.[7]
Medical uses[edit]
In the United States, trifarotene isindicatedfor the topical treatment of acne vulgaris in people nine years of age and older.[6]In both Canada and Australia, it is indicated for the topical treatment of acne vulgaris of the face and/or the trunk in people twelve years of age and older.[1][2][3]
Society and culture[edit]
Legal status[edit]
Trifarotene was approved for medical use in the United States in October 2019,[14]in Canada in November 2019,[3]and in Australia in January 2021.[1][2]
References[edit]
- ^abcd"Trifarotene Product Information".Therapeutic Goods Administration(TGA).Archivedfrom the original on 23 May 2021.Retrieved23 May2021.
- ^abcd"Aklief".Therapeutic Goods Administration (TGA).28 January 2021.Archivedfrom the original on 9 September 2021.Retrieved8 September2021.
- ^abc"PRODUCT MONOGRAPH Trifarotene cream"(PDF).Archived(PDF)from the original on 23 May 2021.Retrieved23 May2021.
- ^"Summary Basis of Decision (SBD) for Aklief".Health Canada.23 October 2014.Archivedfrom the original on 31 May 2022.Retrieved29 May2022.
- ^"Aklief Summary of Product Characteristics (SmPC)".(emc).28 November 2022.Archivedfrom the original on 6 August 2023.Retrieved5 July2024.
- ^abcd"Aklief- trifarotene cream".DailyMed.Archivedfrom the original on 23 May 2021.Retrieved22 May2021.
- ^ab"Galderma receives a positive outcome through the European Decentralised Procedure for Aklief (trifarotene 50 mcg/g cream), the first new retinoid molecule for acne in the European Union in 25 years"(Press release). Galderma. 20 December 2019.Archivedfrom the original on 24 May 2021.Retrieved24 May2021– via Business Wire.
- ^ab"Drug Trials Snapshots: Aklief".U.S.Food and Drug Administration(FDA).11 October 2019.Archivedfrom the original on 19 November 2019.Retrieved18 November2019.
 This article incorporates text from this source, which is in thepublic domain.
This article incorporates text from this source, which is in thepublic domain.
- ^"Trifarotene Monograph for Professionals".Drugs.com.28 October 2019.Archivedfrom the original on 4 March 2021.Retrieved23 May2021.
- ^Scott LJ (November 2019)."Trifarotene: First Approval".Drugs.79(17): 1905–09.doi:10.1007/s40265-019-01218-6.PMID31713811.S2CID207964653.Archivedfrom the original on 29 August 2021.Retrieved17 June2020.
- ^"Trifarotene Orphan Drug Designations and Approvals".U.S.Food and Drug Administration(FDA).24 December 1999.Archivedfrom the original on 29 August 2021.Retrieved19 August2020.
- ^"EU/3/20/2264".European Medicines Agency(EMA).12 August 2020.Archivedfrom the original on 9 January 2021.Retrieved19 August2020.
- ^"Aklief (trifarotene) FDA Approval History".Drugs.com.7 October 2019.Archivedfrom the original on 27 October 2020.Retrieved19 November2019.
- ^ab"Drug Approval Package: Aklief".U.S.Food and Drug Administration(FDA).21 October 2019.Archivedfrom the original on 19 November 2019.Retrieved18 November2019.
