UR-144
 | |
| Clinical data | |
|---|---|
| Trade names | different brandnames, typical example: "TMCP-018", "KM-X1", "UR-144", "MN-001", "YX-17" |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChemCID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard(EPA) | |
| Chemical and physical data | |
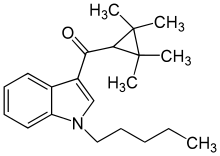
| Formula | C21H29NO |
| Molar mass | 311.469g·mol−1 |
| 3D model (JSmol) | |
| |
| |
UR-144(TMCP-018,KM-X1,MN-001,YX-17) is a drug invented byAbbott Laboratories,[2]that acts as a selectivefull agonistof the peripheralcannabinoid receptorCB2,but with much loweraffinityfor the psychoactiveCB1receptor.
Pharmacology
[edit]UR-144 has high affinity for the CB2receptor with a Kiof 1.8 nM but 83x lower affinity for the CB1receptor with a Kiof 150 nM.[3]UR-144 was found to possess an EC50of 421 nM for human CB1receptors, and 72 nM for human CB2receptors.[4]UR-144 producesbradycardiaand hypothermia in rats at a dose of 10 mg/kg, suggesting weak cannabinoid-like activity.[4]
Chemically it is closely related to other 2,2,3,3-tetramethylcyclopropyl synthetic cannabinoids likeA-796,260andA-834,735but with a different substitution on the 1-position of the indole core, in these compounds its 1-pentyl group is replaced with alkylheterocycles like 1-(2-morpholinoethyl) and 1-(tetrahydropyran-4-ylmethyl).
Legality
[edit]The UK ACMD recommended that generic prohibition legislation be extended to include UR-144 in October 2012.[5]The UK Home Office accepted the recommendation and enacted legislation to ban UR-144 as aclass Bdrug along with a number of other drugs on February 26, 2013 as a part ofThe Misuse of Drugs Act 1971 (Amendment) Order 2013.
UR-144 was detected in Korea, 2012. This molecule is very close to KM-X1, MN-001, YX-17 and Kr-11.[6]
UR-144 (Abbott patent) has been detected as an ingredient ofsynthetic cannabissmoking blends in New Zealand, and subsequently banned from sale as atemporary class drugon 6 April 2012.[7]It has also been encountered in smoking blends and subsequently banned in Russia.[8]
As of October 2015 UR-144 is a controlled substance in China.[9]
UR-144 is banned in the Czech Republic.[10]
Detection
[edit]A forensic standard of UR-144 is available, and the compound has been posted on the Forendex website of potential drugs of abuse.[11]An ELISA immunoassay technique for detecting UR-144 in urine as part of general drug screens has been developed by Tulip Biolabs, Inc. An Homogeneous Immunoassay that runs on most Clinical Chemistry Analyzers and detects several UR and XLR synthetic cannabinoids has been developed and introduced by Immunalysis Inc. Pomona USA.
See also
[edit]- AB-001
- AM-1221
- 4-HTMPIPO
- FAB-144
- JTE 7-31
- JWH-018
- N-(S)-Fenchyl-1-(2-morpholinoethyl)-7-methoxyindole-3-carboxamide
- XLR-11
- Tetramethylcyclopropylfentanyl
References
[edit]- ^Anvisa(2023-07-24)."RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial"[Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese).Diário Oficial da União(published 2023-07-25).Archivedfrom the original on 2023-08-27.Retrieved2023-08-27.
- ^WO application 2006069196,Pace JM, Tietje K, Dart MJ, Meyer MD, "3-Cycloalkylcarbonyl indoles as cannabinoid receptor ligands", published 2006-06-29, assigned to Abbott Laboratories
- ^Frost JM, Dart MJ, Tietje KR, Garrison TR, Grayson GK, Daza AV, et al. (January 2010). "Indol-3-ylcycloalkyl ketones: effects of N1 substituted indole side chain variations on CB(2) cannabinoid receptor activity".Journal of Medicinal Chemistry.53(1): 295–315.doi:10.1021/jm901214q.PMID19921781.
- ^abBanister SD, Stuart J, Kevin RC, Edington A, Longworth M, Wilkinson SM, et al. (August 2015)."Effects of bioisosteric fluorine in synthetic cannabinoid designer drugs JWH-018, AM-2201, UR-144, XLR-11, PB-22, 5F-PB-22, APICA, and STS-135".ACS Chemical Neuroscience.6(8): 1445–1458.doi:10.1021/acschemneuro.5b00107.PMID25921407.
- ^"Further consideration of the synthetic cannabinoids".UK Advisory Council on the Misuse of Drugs. 18 October 2012. p. 14.Retrieved13 December2014.
- ^Almada M, Alves P, Fonseca BM, Carvalho F, Queirós CR, Gaspar H, et al. (February 2020)."Synthetic cannabinoids JWH-018, JWH-122, UR-144 and the phytocannabinoid THC activate apoptosis in placental cells".Toxicology Letters.319:129–137.doi:10.1016/j.toxlet.2019.11.004.PMID31730886.S2CID208061836.
- ^Temporary Class Drug Notices. New Zealand Ministry of Health
- ^Sobolevsky T, Prasolov I, Rodchenkov G (October 2012). "Detection of urinary metabolites of AM-2201 and UR-144, two novel synthetic cannabinoids".Drug Testing and Analysis.4(10): 745–753.doi:10.1002/dta.1418.PMID23042760.
- ^"Quan vu ấn phát 《 phi dược dụng loại ma túy dược phẩm hòa tinh thần dược phẩm liệt quản bạn pháp 》 đích thông tri"(in Chinese). China Food and Drug Administration. 27 September 2015.Retrieved1 October2015.
- ^"Látky, o které byl doplněn seznam č. 4 psychotropních látek (příloha č. 4 k nařízení vlády č. 463/2013 Sb.)"(PDF)(in Czech). Ministerstvo zdravotnictví.
- ^Southern Association of Forensic Scientistshttp://forendex.southernforensic.org/index.php/detail/index/1218
Further reading
[edit]- Poso A, Huffman JW (January 2008)."Targeting the cannabinoid CB2 receptor: modelling and structural determinants of CB2 selective ligands".British Journal of Pharmacology.153(2): 335–346.doi:10.1038/sj.bjp.0707567.PMC2219524.PMID17982473.
- Chin CL, Tovcimak AE, Hradil VP, Seifert TR, Hollingsworth PR, Chandran P, et al. (January 2008)."Differential effects of cannabinoid receptor agonists on regional brain activity using pharmacological MRI".British Journal of Pharmacology.153(2): 367–379.doi:10.1038/sj.bjp.0707506.PMC2219521.PMID17965748.
- Frost JM, Dart MJ, Tietje KR, Garrison TR, Grayson GK, Daza AV, et al. (March 2008). "Indol-3-yl-tetramethylcyclopropyl ketones: effects of indole ring substitution on CB2 cannabinoid receptor activity".Journal of Medicinal Chemistry.51(6): 1904–1912.doi:10.1021/jm7011613.PMID18311894.
