Zeeman effect

TheZeeman effect(/ˈzeɪmən/;Dutch pronunciation:[ˈzeːmɑn]) is the effect of splitting of aspectral lineinto several components in the presence of a staticmagnetic field.It is named after theDutchphysicistPieter Zeeman,who discovered it in 1896 and received a Nobel prize for this discovery. It is analogous to theStark effect,the splitting of a spectral line into several components in the presence of anelectric field.Also similar to the Stark effect, transitions between different components have, in general, different intensities, with some being entirely forbidden (in thedipoleapproximation), as governed by theselection rules.
Since the distance between the Zeeman sub-levels is a function of magnetic field strength, this effect can be used to measure magnetic field strength, e.g. that of theSunand otherstarsor in laboratoryplasmas.
Discovery
[edit]In 1896 Zeeman learned that his laboratory had one ofHenry Augustus Rowland's highest resolvingRowland grating,an imaging spectrographic mirror. Zeeman had readJames Clerk Maxwell's article inEncyclopædia BritannicadescribingMichael Faraday's failed attempts to influence light with magnetism. Zeeman wondered if the new spectrographic techniques could succeed where early efforts had not.[1]: 75
When illuminated by a slit shaped source, the grating produces a long array of slit images corresponding to different wavelengths. Zeeman placed a piece of asbestos soaked in salt water into aBunsen burnerflame at the source of the grating: he could easily see two lines for sodium light emission. Energizing a 10 kilogauss magnet around the flame he observed a slight broadening of the sodium images.[1]: 76
When Zeeman switched tocadmiumat the source he observed the images split when the magnet was energized. These splitting could be analyzed withHendrik Lorentz's then newelectron theory.In retrospect we now know that the magnetic effects on sodium require quantum mechanical treatment.[1]: 77 Zeeman and Lorentz were awarded the 1902 Nobel prize; in his acceptance speech Zeeman explained his apparatus and showed slides of the spectrographic images.[2]
Nomenclature
[edit]Historically, one distinguishes between thenormaland ananomalous Zeeman effect(discovered byThomas Prestonin Dublin, Ireland[3]). The anomalous effect appears on transitions where the netspinof theelectronsis non-zero. It was called "anomalous" because the electron spin had not yet been discovered, and so there was no good explanation for it at the time that Zeeman observed the effect.Wolfgang Paulirecalled that when asked by a colleague as to why he looked unhappy, he replied, "How can one look happy when he is thinking about the anomalous Zeeman effect?"[4]
At higher magnetic field strength the effect ceases to be linear. At even higher field strengths, comparable to the strength of the atom's internal field, the electron coupling is disturbed and the spectral lines rearrange. This is called thePaschen–Back effect.
In the modern scientific literature, these terms are rarely used, with a tendency to use just the "Zeeman effect". Another rarely used obscure term isinverse Zeeman effect,[5]referring to the Zeeman effect in an absorption spectral line.
A similar effect, splitting of thenuclearenergy levels in the presence of a magnetic field, is referred to as thenuclear Zeeman effect.[6]
Theoretical presentation
[edit]The totalHamiltonianof an atom in a magnetic field is
whereis the unperturbed Hamiltonian of the atom, andis theperturbationdue to the magnetic field:
whereis themagnetic momentof the atom. The magnetic moment consists of the electronic and nuclear parts; however, the latter is many orders of magnitude smaller and will be neglected here. Therefore,
whereis theBohr magneton,is the total electronicangular momentum,andis theLandé g-factor. A more accurate approach is to take into account that the operator of the magnetic moment of an electron is a sum of the contributions of theorbital angular momentumand thespin angular momentum,with each multiplied by the appropriategyromagnetic ratio:
whereand(the latter is called theanomalous gyromagnetic ratio;the deviation of the value from 2 is due to the effects ofquantum electrodynamics). In the case of theLS coupling,one can sum over all electrons in the atom:
whereandare the total spin momentum and spin of the atom, and averaging is done over a state with a given value of the total angular momentum.
If the interaction termis small (less than thefine structure), it can be treated as a perturbation; this is the Zeeman effect proper. In the Paschen–Back effect, described below,exceeds theLS couplingsignificantly (but is still small compared to). In ultra-strong magnetic fields, the magnetic-field interaction may exceed,in which case the atom can no longer exist in its normal meaning, and one talks aboutLandau levelsinstead. There are intermediate cases which are more complex than these limit cases.
Weak field (Zeeman effect)
[edit]If thespin–orbit interactiondominates over the effect of the external magnetic field,andare not separately conserved, only the total angular momentumis. The spin and orbital angular momentum vectors can be thought of as precessing about the (fixed) total angular momentum vector.The (time-) "averaged" spin vector is then the projection of the spin onto the direction of:
and for the (time-) "averaged" orbital vector:
Thus,
Usingand squaring both sides, we get
and: usingand squaring both sides, we get
Combining everything and taking,we obtain the magnetic potential energy of the atom in the applied external magnetic field,
where the quantity in square brackets is theLandé g-factorgJof the atom (and) andis the z-component of the total angular momentum. For a single electron above filled shellsand,the Landé g-factor can be simplified into:
Takingto be the perturbation, the Zeeman correction to the energy is
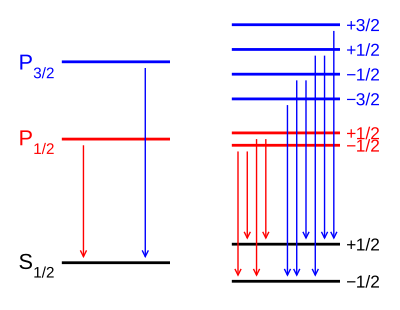
Example: Lyman-alpha transition in hydrogen
[edit]TheLyman-alpha transitioninhydrogenin the presence of thespin–orbit interactioninvolves the transitions
- and
In the presence of an external magnetic field, the weak-field Zeeman effect splits the 1S1/2and 2P1/2levels into 2 states each () and the 2P3/2level into 4 states (). The Landé g-factors for the three levels are:
- for(j=1/2, l=0)
- for(j=1/2, l=1)
- for(j=3/2, l=1).
Note in particular that the size of the energy splitting is different for the different orbitals, because the gJvalues are different. On the left, fine structure splitting is depicted. This splitting occurs even in the absence of a magnetic field, as it is due to spin–orbit coupling. Depicted on the right is the additional Zeeman splitting, which occurs in the presence of magnetic fields.
| Initial state
()
|
Final state
()
|
Energy perturbation |
|---|---|---|
Strong field (Paschen–Back effect)
[edit]The Paschen–Back effect is the splitting of atomic energy levels in the presence of a strong magnetic field. This occurs when an external magnetic field is sufficiently strong to disrupt the coupling between orbital () and spin () angular momenta. This effect is the strong-field limit of the Zeeman effect. When,the two effects are equivalent. The effect was named after theGermanphysicistsFriedrich PaschenandErnst E. A. Back.[7]
When the magnetic-field perturbation significantly exceeds the spin–orbit interaction, one can safely assume.This allows the expectation values ofandto be easily evaluated for a state.The energies are simply
The above may be read as implying that the LS-coupling is completely broken by the external field. Howeverandare still "good" quantum numbers. Together with theselection rulesfor anelectric dipole transition,i.e.,this allows to ignore the spin degree of freedom altogether. As a result, only three spectral lines will be visible, corresponding to theselection rule. The splittingisindependentof the unperturbed energies and electronic configurations of the levels being considered.
More precisely, if,each of these three components is actually a group of several transitions due to the residual spin–orbit coupling and relativistic corrections (which are of the same order, known as 'fine structure'). The first-order perturbation theory with these corrections yields the following formula for the hydrogen atom in the Paschen–Back limit:[8]
Example: Lyman-alpha transition in hydrogen
[edit]In this example, the fine-structure corrections are ignored.
| Initial state
()
|
Initial energy perturbation | Final state
()
|
Final energy perturbation |
|---|---|---|---|
Intermediate field for j = 1/2
[edit]In the magnetic dipole approximation, the Hamiltonian which includes both thehyperfineand Zeeman interactions is
whereis the hyperfine splitting (in Hz) at zero applied magnetic field,andare theBohr magnetonandnuclear magnetonrespectively,andare the electron and nuclear angular momentum operators andis theLandé g-factor:
In the case of weak magnetic fields, the Zeeman interaction can be treated as a perturbation to thebasis. In the high field regime, the magnetic field becomes so strong that the Zeeman effect will dominate, and one must use a more complete basis ofor justsinceandwill be constant within a given level.
To get the complete picture, including intermediate field strengths, we must consider eigenstates which are superpositions of theandbasis states. For,the Hamiltonian can be solved analytically, resulting in theBreit–Rabi formula(named afterGregory BreitandIsidor Isaac Rabi). Notably, the electric quadrupole interaction is zero for(), so this formula is fairly accurate.
We now utilize quantum mechanicalladder operators,which are defined for a general angular momentum operatoras
These ladder operators have the property
as long aslies in the range(otherwise, they return zero). Using ladder operatorsand We can rewrite the Hamiltonian as
We can now see that at all times, the total angular momentum projectionwill be conserved. This is because bothandleave states with definiteandunchanged, whileandeither increaseand decreaseor vice versa, so the sum is always unaffected. Furthermore, sincethere are only two possible values ofwhich are.Therefore, for every value ofthere are only two possible states, and we can define them as the basis:
This pair of states is atwo-level quantum mechanical system.Now we can determine the matrix elements of the Hamiltonian:
Solving for the eigenvalues of this matrix – as can be done by hand (seetwo-level quantum mechanical system), or more easily, with a computer algebra system – we arrive at the energy shifts:
whereis the splitting (in units of Hz) between two hyperfine sublevels in the absence of magnetic field, is referred to as the 'field strength parameter' (Note: forthe expression under the square root is an exact square, and so the last term should be replaced by). This equation is known as theBreit–Rabi formulaand is useful for systems with one valence electron in an() level.[9][10]
Note that indexinshould be considered not as total angular momentum of the atom but asasymptotic total angular momentum.It is equal to total angular momentum only if otherwise eigenvectors corresponding different eigenvalues of the Hamiltonian are the superpositions of states with differentbut equal(the only exceptions are).
Applications
[edit]Astrophysics
[edit]
George Ellery Halewas the first to notice the Zeeman effect in the solar spectra, indicating the existence of strong magnetic fields in sunspots. Such fields can be quite high, on the order of 0.1teslaor higher. Today, the Zeeman effect is used to producemagnetogramsshowing the variation of magnetic field on the Sun.[citation needed]
Laser cooling
[edit]The Zeeman effect is utilized in manylaser coolingapplications such as amagneto-optical trapand theZeeman slower.[citation needed]
Spintronics
[edit]Zeeman-energy mediated coupling of spin and orbital motions is used inspintronicsfor controlling electron spins in quantum dots throughelectric dipole spin resonance.[11]
Metrology
[edit]Old high-precision frequency standards, i.e. hyperfine structure transition-based atomic clocks, may require periodic fine-tuning due to exposure to magnetic fields. This is carried out by measuring the Zeeman effect on specific hyperfine structure transition levels of the source element (cesium) and applying a uniformly precise, low-strength magnetic field to said source, in a process known asdegaussing.[12]
The Zeeman effect may also be utilized to improve accuracy inatomic absorption spectroscopy.[citation needed]
Biology
[edit]A theory about themagnetic senseof birds assumes that a protein in the retina is changed due to the Zeeman effect.[13]
Nuclear spectroscopy
[edit]The nuclear Zeeman effect is important in such applications asnuclear magnetic resonancespectroscopy,magnetic resonance imaging(MRI), andMössbauer spectroscopy.[citation needed]
Other
[edit]Theelectron spin resonancespectroscopy is based on the Zeeman effect.[citation needed]
Demonstrations
[edit]
The Zeeman effect can be demonstrated by placing a sodium vapor source in a powerful electromagnet and viewing a sodium vapor lamp through the magnet opening (see diagram). With magnet off, the sodium vapor source will block the lamp light; when the magnet is turned on the lamp light will be visible through the vapor.
The sodium vapor can be created by sealing sodium metal in an evacuated glass tube and heating it while the tube is in the magnet.[14]
Alternatively, salt (sodium chloride) on a ceramic stick can be placed in the flame ofBunsen burneras the sodium vapor source. When the magnetic field is energized, the lamp image will be brighter.[15]However, the magnetic field also affects the flame, making the observation depend upon more than just the Zeeman effect.[14]These issues also plagued Zeeman's original work; he devoted considerable effort to ensure his observations were truly an effect of magnetism on light emission.[16]
When salt is added to the Bunsen burner, itdissociatesto givesodiumandchloride.The sodium atoms get excited due tophotonsfrom the sodium vapour lamp, with electrons excited from 3s to 3p states, absorbing light in the process. The sodium vapour lamp emits light at 589nm, which has precisely the energy to excite an electron of a sodium atom. If it was an atom of another element, like chlorine, shadow will not be formed.[17][failed verification]When a magnetic field is applied, due to the Zeeman effect thespectral lineof sodium gets split into several components. This means the energy difference between the 3s and 3patomic orbitalswill change. As the sodium vapour lamp don't precisely deliver the right frequency any more, light doesn't get absorbed and passes through, resulting in the shadow dimming. As the magnetic field strength is increased, the shift in the spectral lines increases and lamp light is transmitted.[citation needed]
See also
[edit]- Magneto-optic Kerr effect
- Voigt effect
- Faraday effect
- Cotton–Mouton effect
- Polarization spectroscopy
- Quantum light dimmer
- Zeeman energy
- Stark effect
- Lamb shift
References
[edit]- ^abcPais, Abraham (2002).Inward bound: of matter and forces in the physical world(Reprint ed.). Oxford: Clarendon Press [u.a.]ISBN978-0-19-851997-3.
- ^Pieter, Zeeman (1902)."Pieter Zeeman Nobel Lecture".The Nobel Prize.Archivedfrom the original on 15 November 2018.Retrieved1 March2024.
- ^Preston, Thomas (1898)."Radiation phenomena in a strong magnetic field".The Scientific Transactions of the Royal Dublin Society.2nd series.6:385–391.
- ^"Niels Bohr's Times: In Physics, Philosophy, and Polity" By Abraham Pais, page 201
- ^Jenkins, Francis; White, Harvey (3 December 2001).Fundamentals of Optics(4 ed.). McGraw-Hill Education.ISBN978-0-07-256191-3.
- ^Dunlap, Richard A. (1 December 2023)."Hyperfine interactions—part III: the magnetic dipole interaction and the nuclear Zeeman effect".The Mössbauer Effect (Second Edition).doi:10.1088/978-0-7503-6039-5ch7.ISBN978-0-7503-6039-5.Retrieved4 March2024.
- ^Paschen, F.; Back, E. (1921). "Liniengruppen magnetisch vervollständigt" [Line groups magnetically completed [i.e., completely resolved]].Physica(in German).1:261–273.Available at:Leiden University (Netherlands)
- ^Griffiths, David J. (2004).Introduction to Quantum Mechanics(2nd ed.).Prentice Hall.p. 280.ISBN0-13-111892-7.OCLC40251748.
- ^Woodgate, Gordon Kemble (1980).Elementary Atomic Structure(2nd ed.). Oxford, England: Oxford University Press. pp. 193–194.
- ^First appeared in:Breit, G.; Rabi, I.I. (1931). "Measurement of nuclear spin".Physical Review.38(11): 2082–2083.Bibcode:1931PhRv...38.2082B.doi:10.1103/PhysRev.38.2082.2.
- ^Y. Tokura, W. G. van der Wiel, T. Obata, and S. Tarucha, Coherent single electron spin control in a slanting Zeeman field, Phys. Rev. Lett.96,047202 (2006)
- ^Verdiell, Marc (CuriousMarc) (31 October 2022).How an Atomic Clock Really Works, Round 2: Zeeman Alignment(YouTube video).Retrieved11 March2023.
- ^Thalau, Peter; Ritz, Thorsten; Burda, Hynek; Wegner, Regina E.; Wiltschko, Roswitha (18 April 2006)."The magnetic compass mechanisms of birds and rodents are based on different physical principles".Journal of the Royal Society Interface.3(9): 583–587.doi:10.1098/rsif.2006.0130.PMC1664646.PMID16849254.
- ^abCandle flame is repelled by magnets (and Zeeman follow-up),retrieved27 February2024
- ^Candle flame is repelled by magnets (and Zeeman follow-up),retrieved27 February2024
- ^Kox, A J (1 May 1997)."The discovery of the electron: II. The Zeeman effect".European Journal of Physics.18(3): 139–144.Bibcode:1997EJPh...18..139K.doi:10.1088/0143-0807/18/3/003.ISSN0143-0807.S2CID53414643.
- ^Suzuki, Masatsugu Sei; Suzuki, Itsuko S. (2011)."Lecture Note on Senior Laboratory Zeeman effect in Na, Cd, and Hg".ResearchGate.
Historical
[edit]- Condon, E. U.; G. H. Shortley (1935).The Theory of Atomic Spectra.Cambridge University Press.ISBN0-521-09209-4.(Chapter 16 provides a comprehensive treatment, as of 1935.)
- Zeeman, P. (1896)."Over de invloed eener magnetisatie op den aard van het door een stof uitgezonden licht"[On the influence of magnetism on the nature of the light emitted by a substance].Verslagen van de Gewone Vergaderingen der Wis- en Natuurkundige Afdeeling (Koninklijk Akademie van Wetenschappen te Amsterdam) [Reports of the Ordinary Sessions of the Mathematical and Physical Section (Royal Academy of Sciences in Amsterdam)](in Dutch).5:181–184 and 242–248.Bibcode:1896VMKAN...5..181Z.
- Zeeman, P. (1897)."On the influence of magnetism on the nature of the light emitted by a substance".Philosophical Magazine.5th series.43(262): 226–239.doi:10.1080/14786449708620985.
- Zeeman, P. (11 February 1897)."The effect of magnetisation on the nature of light emitted by a substance".Nature.55(1424): 347.Bibcode:1897Natur..55..347Z.doi:10.1038/055347a0.
- Zeeman, P. (1897)."Over doubletten en tripletten in het spectrum, teweeggebracht door uitwendige magnetische krachten"[On doublets and triplets in the spectrum, caused by external magnetic forces].Verslagen van de Gewone Vergaderingen der Wis- en Natuurkundige Afdeeling (Koninklijk Akademie van Wetenschappen te Amsterdam) [Reports of the Ordinary Sessions of the Mathematical and Physical Section (Royal Academy of Sciences in Amsterdam)](in Dutch).6:13–18, 99–102, and 260–262.
- Zeeman, P. (1897)."Doublets and triplets in the spectrum produced by external magnetic forces".Philosophical Magazine.5th series.44(266): 55–60.doi:10.1080/14786449708621028.
Modern
[edit]- Feynman, Richard;Leighton, Robert B.;Sands, Matthew(1989).The Feynman Lectures on Physics.Vol. 3.Addison-Wesley.ISBN0-201-02115-3.
- Forman, Paul (1970). "Alfred Landé and the anomalous Zeeman Effect, 1919-1921".Historical Studies in the Physical Sciences.2:153–261.doi:10.2307/27757307.JSTOR27757307.
- Griffiths, David J. (2004).Introduction to Quantum Mechanics(2nd ed.).Prentice Hall.ISBN0-13-805326-X.
- Liboff, Richard L.(2002).Introductory Quantum Mechanics(4th ed.).Addison-Wesley.ISBN0-8053-8714-5.OCLC50475492.
- Sobelman, Igor I. (2006).Theory of Atomic Spectra.Alpha Science.ISBN1-84265-203-6.OCLC71825022.
- Foot, C.J. (2005).Atomic Physics.OUP Oxford.ISBN0-19-850696-1.OCLC57478010.






















![{\displaystyle {\vec {S}}\cdot {\vec {J}}={\frac {1}{2}}(J^{2}+S^{2}-L^{2})={\frac {\hbar ^{2}}{2}}[j(j+1)-l(l+1)+s(s+1)],}](https://wikimedia.org/api/rest_v1/media/math/render/svg/26e9f5a54c934ff91a0ee2e1e0711656fc8a5110)

![{\displaystyle {\vec {L}}\cdot {\vec {J}}={\frac {1}{2}}(J^{2}-S^{2}+L^{2})={\frac {\hbar ^{2}}{2}}[j(j+1)+l(l+1)-s(s+1)].}](https://wikimedia.org/api/rest_v1/media/math/render/svg/394b91804fd17c811c0003f1d0ea096d640f39b7)

![{\displaystyle {\begin{aligned}V_{\rm {M}}&=\mu _{\rm {B}}Bm_{j}\left[g_{L}{\frac {j(j+1)+l(l+1)-s(s+1)}{2j(j+1)}}+g_{S}{\frac {j(j+1)-l(l+1)+s(s+1)}{2j(j+1)}}\right]\\&=\mu _{\rm {B}}Bm_{j}\left[1+(g_{S}-1){\frac {j(j+1)-l(l+1)+s(s+1)}{2j(j+1)}}\right],\\&=\mu _{\rm {B}}Bm_{j}g_{j}\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/213add165a96705cd8e71d5cae67f97afa873ced)































![{\displaystyle [H_{0},S]=0}](https://wikimedia.org/api/rest_v1/media/math/render/svg/c381cfc71af43948523f8c7d1a9fc9d2c8246665)










![{\displaystyle E_{z+fs}=E_{z}+{\frac {m_{e}c^{2}\alpha ^{4}}{2n^{3}}}\left\{{\frac {3}{4n}}-\left[{\frac {l(l+1)-m_{l}m_{s}}{l(l+1/2)(l+1)}}\right]\right\}.}](https://wikimedia.org/api/rest_v1/media/math/render/svg/ba7927da53bd8a30b0213965a2a7cad81d83e9c5)


























































