Aliphatic compound
This articlerelies largely or entirely on asingle source.(October 2022) |


Inorganic chemistry,hydrocarbons(compoundscomposed solely ofcarbonandhydrogen) are divided into two classes:aromatic compoundsandaliphatic compounds(/ˌælɪˈfætɪk/;G.aleiphar,fat, oil). Aliphatic compounds can besaturated(in which all the C-C bonds are single requiring the structure to be completed, or 'saturated', by hydrogen) likehexane,orunsaturated,likehexeneandhexyne.Open-chain compounds,whether straight or branched, and which contain no rings of any type, are always aliphatic.Cyclic compoundscan be aliphatic if they are notaromatic.[1]
Structure[edit]
Aliphatic compounds can besaturated,joined by single bonds (alkanes), or unsaturated, with double bonds (alkenes) or triple bonds (alkynes). If other elements (heteroatoms) are bound to thecarbon chain,the most common beingoxygen,nitrogen,sulfur,andchlorine,it is no longer a hydrocarbon, and therefore no longer an aliphatic compound. However, such compounds may still be referred to as aliphatic if the hydrocarbon portion of the molecule is aliphatic, e.g.aliphatic amines,to differentiate them fromaromatic amines.
The least complex aliphatic compound ismethane(CH4).
Properties[edit]
Most aliphatic compounds areflammable,allowing the use ofhydrocarbonsasfuel,such as methane innatural gasfor stoves or heating;butaneintorchesandlighters;various aliphatic (as well as aromatic) hydrocarbons in liquid transportation fuels likepetrol/gasoline,diesel,andjet fuel;and other uses such asethyne(acetylene) inwelding.
Examples of aliphatic compounds[edit]
The most important aliphatic compounds are:
- n-, iso- and cyclo-alkanes (saturated hydrocarbons)
- n-, iso- and cyclo-alkenes and -alkynes (unsaturated hydrocarbons).
Important examples of low-molecular aliphatic compounds can be found in the list below (sorted by the number of carbon-atoms):
| Formula | Name | Structural formula | Chemical classification |
|---|---|---|---|
| CH4 | Methane | 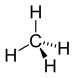 |
Alkane |
| C2H2 | Acetylene | Alkyne | |
| C2H4 | Ethylene | 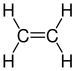 |
Alkene |
| C2H6 | Ethane |  |
Alkane |
| C3H4 | Propyne | Alkyne | |
| C3H6 | Propene | 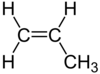 |
Alkene |
| C3H8 | Propane |  |
Alkane |
| C4H6 | 1,2-Butadiene |  |
Diene |
| C4H6 | 1-Butyne |  |
Alkyne |
| C4H8 | 1-Butene | Alkene | |
| C4H10 | Butane | 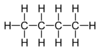 |
Alkane |
| C6H10 | Cyclohexene | Cycloalkene | |
| C5H12 | n-pentane | Alkane | |
| C7H14 | Cycloheptane | Cycloalkane | |
| C7H14 | Methylcyclohexane | 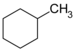 |
Cyclohexane |
| C8H8 | Cubane |  |
Prismane,Platonic hydrocarbon |
| C9H20 | Nonane | Alkane | |
| C10H12 | Dicyclopentadiene |  |
Diene, Cycloalkene |
| C10H16 | Phellandrene |   |
Terpene,Diene, Cycloalkene |
| C10H16 | α-Terpinene | Terpene, Diene, Cycloalkene | |
| C10H16 | Limonene |  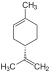 |
Terpene, Diene, Cycloalkene |
| C11H24 | Undecane | Alkane | |
| C30H50 | Squalene | Terpene, Polyene | |
| C2nH4n | Polyethylene | Alkane |
References[edit]
- ^IUPAC,Compendium of Chemical Terminology,2nd ed. (the "Gold Book" ) (1997). Online corrected version: (1995) "aliphatic compounds".doi:10.1351/goldbook.A00217
