Apronal
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code | |
| Pharmacokineticdata | |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChemCID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard(EPA) | |
| ECHA InfoCard | 100.007.677 |
| Chemical and physical data | |
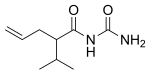
| Formula | C9H16N2O2 |
| Molar mass | 184.239g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Apronal(brand nameSedormid), orapronalide,also known asallylisopropylacetylureaorallylisopropylacetylcarbamide,is ahypnotic/sedativedrugof theureide(acylurea) group synthesized in 1926[1]byHoffmann-La Roche.Though it is not abarbiturate,apronalide is similar instructureto the barbiturates (being an open-chaincarbamideinstead of having aheterocyclic ring).[2]In accordance, it is similar in action to the barbiturates, although considerably milder in comparison (formerly used as a daytime sedative at doses of 1 to 2 grams every 3 to 4 hours).[2]Upon the finding that it caused patients to developthrombocytopenic purpura,apronalide was withdrawn from clinical use.[3]
Medicines with allylisopropylacetylurea are no longer used except inJapan.[3]Notably AustralianTherapeutic Goods Administrationissued a safety alert in May 2023 which prohibits the sale, supply and use of Japanese EVE-branded products in Australia[4]due to its dangerous side effects.
See also[edit]
References[edit]
- ^DE 459903,"Verfahren zur Darstellung von Ureiden der Dialkylessigsaeuren", issued 15 May 1928, assigned to Hoffmann-La Roche
- ^abRoche Review...Hoffman-La Roche, and Roche-organon. 1938. p. 164.
- ^abVollum RL, Jamison DG, Cummins CS (20 May 2014).Fairbrother's Textbook of Bacteriology.Elsevier Science. pp. 152–.ISBN978-1-4831-4178-7.
- ^"EVE Allylisopropylacetylurea tablets".Therapeutic Goods Administration (TGA).Retrieved31 May2023.
