Detergent

Adetergentis asurfactantor amixtureof surfactants withcleansingproperties when indilutesolutions.[1]There are a large variety of detergents, a common family being thealkylbenzene sulfonates,which aresoap-like compounds that are more soluble inhard water,because the polarsulfonate(of detergents) is less likely than the polarcarboxylate(of soap) to bind to calcium and other ions found in hard water.
Definitions
[edit]The worddetergentis derived from the Latin adjectivedetergens,from the verbdetergere,meaning to wipe or polish off. Detergent can be defined as asurfactantor amixtureof surfactants with cleansing properties when indilutesolutions.[1]However, conventionally, detergent is used to mean synthetic cleaning compounds as opposed tosoap(a salt of the naturalfatty acid), even though soap is also a detergent in the true sense.[2]In domestic contexts, the termdetergentrefers to household cleaning products such aslaundry detergentordish detergent,which are in fact complex mixtures of different compounds, not all of which are by themselves detergents.
Detergency is the ability to remove unwanted substances termed 'soils' from a substrate (e.g., clothing).[3]
Structure and properties
[edit]Detergents are a group of compounds with an amphiphilic structure, where each molecule has a hydrophilic (polar) head and a long hydrophobic (non-polar) tail. The hydrophobic portion of these molecules may be straight- or branched-chainhydrocarbons,or it may have asteroidstructure. The hydrophilic portion is more varied, they may be ionic or non-ionic, and can range from a simple or a relatively elaborate structure.[4]Detergents are surfactants since they can decrease thesurface tensionof water. Their dual nature facilitates the mixture of hydrophobic compounds (like oil and grease) with water. Because air is not hydrophilic, detergents are alsofoaming agentsto varying degrees.
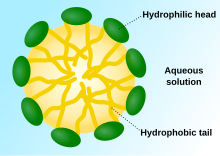
Detergent molecules aggregate to formmicelles,which makes them soluble in water. The hydrophobic group of the detergent is the main driving force of micelle formation, its aggregation forms the hydrophobic core of the micelles. The micelle can remove grease, protein or soiling particles. The concentration at which micelles start to form is thecritical micelle concentration(CMC), and the temperature at which the micelles further aggregate to separate the solution into two phases is the cloud point when the solution becomes cloudy and detergency is optimal.[4]
Detergents work better in an alkaline pH. The properties of detergents are dependent on the molecular structure of themonomer.The ability to foam may be determined by the head group, for example anionic surfactants are high-foaming, while nonionic surfactants may be non-foaming or low-foaming.[5]
Chemical classifications of detergents
[edit]Detergents are classified into four broad groupings, depending on the electrical charge of the surfactants.[6]
Anionic detergents
[edit]Typical anionic detergents arealkylbenzene sulfonates.Thealkylbenzeneportion of theseanionsis lipophilic and the sulfonate is hydrophilic. Two varieties have been popularized, those with branchedalkyl groupsand those with linear alkyl groups. The former were largely phased out in economically advanced societies because they are poorly biodegradable.[7]
Anionic detergents is the most common form of detergents, and an estimated 6 billion kilograms of anionic detergents are produced annually for the domestic markets.
Bile acids,such asdeoxycholic acid(DOC), are anionic detergents produced by the liver to aid in digestion and absorption of fats and oils.

Cationic detergents
[edit]Cationic detergents are similar to anionic ones, butquaternary ammoniumreplaces the hydrophilic anionic sulfonate group. The ammonium sulfate center is positively charged.[7]Cationic surfactants generally have poor detergency.
Non-ionic detergents
[edit]Non-ionic detergents are characterized by their uncharged, hydrophilic headgroups. Typical non-ionic detergents are based onpolyoxyethyleneor aglycoside.Common examples of the former includeTween,Triton,and the Brij series. These materials are also known as ethoxylates or PEGylates and their metabolites,nonylphenol.Glycosides have a sugar as their uncharged hydrophilic headgroup. Examples includeoctyl thioglucosideandmaltosides.HEGA and MEGA series detergents are similar, possessing a sugar alcohol as headgroup.
Amphoteric detergents
[edit]Amphotericor zwitterionic detergents havezwitterionswithin a particular pH range, and possess a net zero charge arising from the presence of equal numbers of +1 and −1 charged chemical groups. Examples includeCHAPS.
History
[edit]Soap is known to have been used as a surfactant for washing clothes since theSumeriantime in 2,500 B.C.[8]Inancient Egypt,sodawas used as a wash additive. In the 19th century, synthetic surfactants began to be created, for example from olive oil.[9]Sodium silicate(water glass) was used in soap-making in the United States in the 1860s,[10]and in 1876,Henkelsold a sodium silicate-based product that can be used with soap and marketed as a "universal detergent" (Universalwaschmittel) in Germany. Soda was then mixed with sodium silicate to produce Germany's first brand name detergent Bleichsoda.[11]In 1907, Henkel also added a bleaching agentsodium perborateto launch the first 'self-acting' laundry detergentPersilto eliminate the laborious rubbing of laundry by hand.[12]
During theFirst World War,there was a shortage of oils and fats needed to make soap. In order to find alternatives for soap, synthetic detergents were made in Germany by chemists using raw material derived from coal tar.[13][14][9]These early products, however, did not provide sufficient detergency. In 1928, effective detergent was made through thesulfationoffatty alcohol,but large-scale production was not feasible until low-cost fatty alcohols become available in the early 1930s.[15]The synthetic detergent created was more effective and less likely to form scum than soap in hard water, and can also eliminate acid and alkaline reactions and decompose dirt. Commercial detergent products with fatty alcohol sulphates began to be sold, initially in 1932 in Germany byHenkel.[15]In the United States, detergents were sold in 1933 byProcter & Gamble(Dreft) primarily in areas with hard water.[14]However, sales in the US grew slowly until the introduction of 'built' detergents with the addition of effectivephosphate builderdeveloped in the early 1940s.[14]The builder improves the performance of the surfactants by softening the water through thechelationof calcium and magnesium ions, helping to maintain an alkaline pH, as well as dispersing and keeping the soiling particles in solution.[16]The development of the petrochemical industry after the Second World War also yielded material for the production of a range of synthetic surfactants, and alkylbenzene sulfonates became the most important detergent surfactants used.[17]By the 1950s,laundry detergentshad become widespread, and largely replaced soap for cleaning clothes in developed countries.[15]
Over the years, many types of detergents have been developed for a variety of purposes, for example, low-sudsing detergents for use in front-loading washing machines, heavy-duty detergents effective in removing grease and dirt, all-purpose detergents and specialty detergents.[14][18]They become incorporated in various products outside of laundry use, for example indishwasher detergents,shampoo, toothpaste, industrial cleaners, and in lubricants and fuels to reduce or prevent the formation of sludge or deposits.[19]The formulation of detergent products may includebleach,fragrances, dyes and other additives. The use ofphosphates in detergent,however, led to concerns overnutrient pollutionand demand for changes to the formulation of the detergents.[20]Concerns were also raised over the use of surfactants such as branched alkylbenzene sulfonate (tetrapropylenebenzene sulfonate) that lingers in the environment, which led to their replacement by surfactants that are more biodegradable, such as linear alkylbenzene sulfonate.[15][17]Developments over the years have included the use ofenzymes,substitutes for phosphates such aszeoliteA andNTA,TAEDasbleach activator,sugar-based surfactants which are biodegradable and milder to skin, and othergreen friendlyproducts, as well as changes to the form of delivery such as tablets, gels andpods.[21][22]
Major applications of detergents
[edit]
Household cleaning
[edit]One of the largest applications of detergents is for household and shop cleaning includingdish washingand washinglaundry.These detergents are commonly available as powders or concentrated solutions, and the formulations of these detergents are often complex mixtures of a variety of chemicals aside from surfactants, reflecting the diverse demands of the application and the highly competitive consumer market. These detergents may contain the following components:[21]
- surfactants
- foam regulators
- builders
- bleach
- bleach activators
- enzymes
- dyes
- fragrances
- other additives
Fuel additives
[edit]Both carburetors and fuel injector components ofinternal combustion enginesbenefit from detergents in the fuels to preventfouling.Concentrations are about 300ppm.Typical detergents are long-chainaminesandamidessuch aspolyisobuteneamineand polyisobuteneamide/succinimide.[23]
Biological reagent
[edit]Reagent gradedetergents are employed for the isolation and purification ofintegral membrane proteinsfound inbiological cells.[24]Solubilization ofcell membranebilayers requires a detergent that can enter the inner membranemonolayer.[25]Advancements in the purity and sophistication of detergents have facilitated structural and biophysical characterization of important membrane proteins such asion channelsalso the disrupt membrane by bindinglipopolysaccharide,[26]transporters,signaling receptors,andphotosystem II.[27]
See also
[edit]- Cleavable detergent
- Dishwashing liquid
- Dispersant
- Green cleaning
- Hard-surface cleaner
- Laundry detergent
- List of cleaning products
- Triton X-100
References
[edit]- ^abIUPAC,Compendium of Chemical Terminology,2nd ed. (the "Gold Book" ) (1997). Online corrected version: (2006–) "detergent".doi:10.1351/goldbook.D01643
- ^NIIR Board of Consultants Engineers (2013).The Complete Technology Book on Detergents(2nd Revised ed.). Niir Project Consultancy Services. p. 1.ISBN9789381039199– via Google Books.
- ^Arno Cahn, ed. (2003).5th World Conference on Detergents.The American Oil Chemists Society. p. 154.ISBN9781893997400– via Google Books.
- ^abNeugebauer, Judith M. (1990). "[18] Detergents: An overview".Detergents: An overview.Methods in Enzymology. Vol. 182. pp. 239–253.doi:10.1016/0076-6879(90)82020-3.ISBN9780121820831.PMID2314239.
- ^Niir Board (1999).Handbook on Soaps, Detergents & Acid Slurry(3rd Revised ed.). Asia Pacific Business Press. p. 270.ISBN9788178330938– via Google Books.
- ^Mehreteab, Ammanuel (1999). Guy Broze (ed.).Handbook of Detergents, Part A.Taylor & Francis. pp. 133–134.ISBN9781439833322– via Google Books.
- ^abEduard Smulders, Wolfgang Rybinski, Eric Sung, Wilfried Rähse, Josef Steber, Frederike Wiebel, Anette Nordskog, "Laundry Detergents" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim.doi:10.1002/14356007.a08_315.pub2
- ^Jürgen Falbe, ed. (2012).Surfactants in Consumer Products.Springer-Verlag. pp. 1–2.ISBN9783642715457– via Google Books.
- ^abPaul Sosis, Uri Zoller, ed. (2008).Handbook of Detergents, Part F.CRC Press. p. 5.ISBN9781420014655.
- ^Aftalion, Fred (2001).A History of the International Chemical Industry.Chemical Heritage Press. p. 82.ISBN9780941901291.
- ^Ward, James; Löhr (2020).The Perfection of the Paper Clip.Atria Books. p. 190.ISBN9781476799872.
- ^Jakobi, Günter; Löhr, Albrecht (2012).Detergents and Textile Washing.Springer-Verlag. pp. 3–4.ISBN9780895736864.
- ^"Soaps & Detergent: History (1900s to Now)".American Cleaning Institute.Retrieved on 6 January 2015
- ^abcdDavid O. Whitten; Bessie Emrick Whitten (1 January 1997).Handbook of American Business History: Extractives, manufacturing, and services.Greenwood Publishing Group. pp. 221–222.ISBN978-0-313-25199-3– via Google Books.
- ^abcdJürgen Falbe, ed. (2012).Surfactants in Consumer Products.Springer-Verlag. pp. 3–5.ISBN9783642715457– via Google Books.
- ^Urban, David G. (2003).How to Formulate and Compound Industrial Detergents.David G. Urban. pp. 4–5.ISBN9781588988683.
- ^abPaul Sosis, Uri Zoller, ed. (2008).Handbook of Detergents, Part F.CRC Press. p. 6.ISBN9781420014655.
- ^Paul Sosis, Uri Zoller, ed. (2008).Handbook of Detergents, Part F.p. 497.ISBN9781420014655.
- ^Uri Zoller, ed. (2008).Handbook of Detergents, Part E: Applications.Taylor & Francis. p. 331.ISBN9781574447576.
- ^David O. Whitten; Bessie Emrick Whitten (1999).Handbook of Detergents, Part A.Taylor & Francis. p. 3.ISBN9781439833322– via Google Books.
- ^abMiddelhauve, Birgit (2003). Arno Cahn (ed.).5th World Conference on Detergents.The American Oil Chemists Society. pp. 64–67.ISBN9781893997400.
- ^Long, Heather."Laundry Detergent History".Love to Know.
- ^Werner Dabelstein, Arno Reglitzky, Andrea Schütze, Klaus Reders "Automotive Fuels" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheimdoi:10.1002/14356007.a16_719.pub2
- ^Koley D, Bard AJ (2010)."Triton X-100 concentration effects on membrane permeability of a single HeLa cell by scanning electrochemical microscopy (SECM)".Proceedings of the National Academy of Sciences of the United States of America.107(39): 16783–7.Bibcode:2010PNAS..10716783K.doi:10.1073/pnas.1011614107.PMC2947864.PMID20837548.
- ^Lichtenberg D, Ahyayauch H, Goñi FM (2013)."The mechanism of detergent solubilization of lipid bilayers".Biophysical Journal.105(2): 289–299.Bibcode:2013BpJ...105..289L.doi:10.1016/j.bpj.2013.06.007.PMC3714928.PMID23870250.
- ^Doyle, DA; Morais Cabral, J; Pfuetzner, RA; Kuo, A; Gulbis, JM; Cohen, SL; Chait, BT; MacKinnon, R (1998). "The structure of the potassium channel: molecular basis of K+conduction and selectivity".Science.280(5360): 69–77.Bibcode:1998Sci...280...69D.doi:10.1126/science.280.5360.69.PMID9525859.
- ^Umena, Yasufumi; Kawakami, Keisuke; Shen, Jian-Ren; Kamiya, Nobuo (2011)."Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9 A"(PDF).Nature.473(7345): 55–60.Bibcode:2011Natur.473...55U.doi:10.1038/nature09913.PMID21499260.S2CID205224374.
External links
[edit]- About: How Do Detergents CleanArchived6 January 2011 at theWayback Machine
- Campbell tipsfor detergents chemistry,surfactants,andhistoryrelated to laundry washing,destaining methodsandsoil.
- Formulation of Detergent
