Xylose isomerase
| Xylose isomerase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 D-Xylose isomerase tetramer fromStreptomyces rubiginosusPDB2glk.[1]One monomer is coloured by secondary structure to highlight the TIM barrel architecture. | |||||||||
| Identifiers | |||||||||
| EC no. | 5.3.1.5 | ||||||||
| CAS no. | 9023-82-9 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDBstructures | RCSB PDBPDBePDBsum | ||||||||
| Gene Ontology | AmiGO/QuickGO | ||||||||
| |||||||||
Inenzymology,axylose isomerase(EC5.3.1.5) is anenzymethatcatalyzesthe interconversion of D-xyloseandD-xylulose.This enzyme belongs to the family ofisomerases,specifically those intramolecularoxidoreductasesinterconvertingaldosesandketoses.The isomerase has now been observed in nearly a hundred species of bacteria.[2]Xylose-isomerases are also commonly called glucose isomerase or fructose isomerases due to their ability to interconvert glucose and fructose. Thesystematic nameof this enzyme class is α-D-xylopyranose aldose-ketose-isomerase. Other names in common use include D-xylose isomerase, D-xylose ketoisomerase, and D-xylose ketol-isomerase.[3]
History
[edit]The activity ofD-xyloseisomerasewas first observed by Mitsuhashi and Lampen in 1953 in the bacteriumLactobacillus pentosus.[4]It has also been successfully produced intransformedE.coli.[5]In 1957, the D-xylose isomerase activity on D-glucose conversion to D-fructose was noted by Kooi and Marshall.[6]It is now known that isomerases have broad substrate specificity. Most pentoses and some hexoses are all substrates for D-xylose isomerase. Some examples include D-ribose, L-arabinose, L-rhamnose, and D-allose.[7]
Conversion of glucose to fructose by xylose isomerase was first patented in the 1960s. However, the process was not industrially practical as the enzymes were in solution, and recycling the enzyme was problematic.[7]An immobile xylose isomerase that was fixed on a solid surface was first developed in Japan by Takanashi.[7]These developments were essential to the development ofindustrial fermentationprocesses used in manufacturinghigh-fructose corn syrup.[8]: 27 [9]: 808–813 [10]
The tertiary structures of several microbial xylose isomerases were determined from the mid 1980s (Streptomyces olivochromogenesin 1988,Streptomyces violaceonigerin 1988,Streptomyces rubiginosusin 1984,Arthrobacter B3728in 1986,Actinoplanes missouriensisin 1992, andClostridium thermosulfurogenesin 1990).[8]: 366
Function
[edit]This enzyme participates inpentose and glucuronate interconversionsandfructoseandmannosemetabolism. The most bio-available sugars according to the International Society of Rare Sugars are: glucose, galactose, mannose, fructose, xylose, ribose, and L-arabinose. Twenty hexoses and nine pentoses, including xylulose, were considered to be "rare sugars". Hence D-xylose isomerase is used to produce these rare sugars which have very important applications in biology despite their low abundance.[11]
Characterization
[edit]Xylose isomerase can be isolated from red Chinese rice wine, which contains the bacteriumLactobacillus xylosus.[12]This bacterium was mistakenly classified as aL. plantarum,which normally grows on the sugarL-arabinose,and rarely grown on D-xylose.L. xylosuswas recognized to be distinct for its ability to grow on D-xylose.[13] Xylose isomerase inL. xylosushas a molecular weight of about 183000 daltons.[14] Its optimum growth pH is about 7.5 for theL. lactis,however strains such as theL.brevisxylose enzyme prefer a more alkaline environment. TheL. lactisstrain is stable over the pH range of 6.5 to 11.0, and theL. brevisenzyme, which is less tolerant of pH changes, show activity over the pH range of 5.7–7.0.[14]Thermal tests were also done by Kei Y. and Noritaka T. and the xylose isomerase was found to be thermally stable to about 60 degrees Celsius[14]
Active site and mechanism
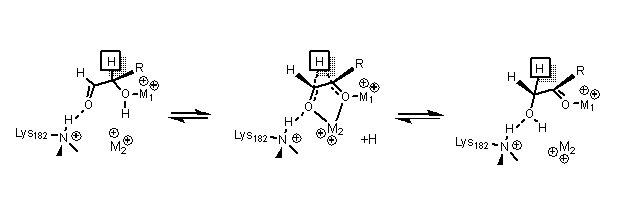
[edit]Xylose isomerase has a structure that is based on eight Alpha /beta barrels that create an active site holding two divalent magnesium ions. Xylose isomerase enzymes exhibit aTIM barrelfold with the active site in the centre of the barrel and atetramericquaternary structure.[15]PDBstructures are available in the links in the infobox to the right. The protein is atetramerwhere paired barrels are nearly coaxial, which form two cavities in which the divalent metals are both bound to one of the two cavities. The metals are in an octahedral geometry. Metal site 1 binds the substrate tightly, while metal site 2 binds the substrate loosely. Both share an acid residue, Glutamic acid 216 of the enzyme, that bridges the two cations. Two basic amino acids surround the negatively chargedligandsto neutralize them. The second cavity faces the metal cavity and both cavities share the same access route. The second cavity is hydrophobic, and has ahistidineresidue activated by anaspartateresidue that ishydrogen bondedto it. This histidine residue is important in theisomerizationof glucose.[16]
In the isomerization of glucose, Histidine 53 is used to catalyze the proton transfer of O1 to O5; the diagram for the ring opening mechanism is shown below. The first metal, mentioned earlier,coordinatesto O3 and O4, and is used to dock the substrate.[16]

In the isomerization of xylose, crystal data shows that xylose binds to the enzyme as an open chain. Metal 1 binds to O2 and O4, and once bound, metal 2 binds to O1 and O2 in the transition state. These interactions along with a lysine residue help catalyze thehydride shiftnecessary for isomerization.[17][16]The transition state consists of a high energycarbonium ionthat is stabilized through all the metal interactions with the sugar substrate.[16]
Application in industry
[edit]The most widely used application of this enzyme is in the conversion of glucose to fructose to producehigh fructose corn syrup(HFCS).[8]: 27 There are three general steps in producing HFCS from starch:[9]: 808–813
- enzymatic degradation of the starch using α-amylase.Also known as liquification.
- further degradation usingglucoamylaseand a debranching enzyme.
- Production of fructose by xylose isomerase
The process is carried out inbioreactorsat 60–65 °C.[8]: 27 Many enzyme becomedenaturedat these temperatures, and one focus of research has been engineering more thermostable versions of xylose isomerase and the other enzymes in the process.[8]: 27 [18]The enzymes are generallyimmobilizedto increase throughput, and finding better ways to do this has been another research focus.[8]: 358–360 [19]
Xylose isomerase is one of the enzymes used by bacteria in nature in order to utilizehemicelluloseas an energy source, and another focus of industrial and academic research has been developing versions of xylose isomerase that could be useful in the production ofbiofuel.[8]: 358 [20][21]
As a dietary supplement
[edit]
The ability of xylose isomerase to convert between glucose and fructose has led to its proposal as a treatment for fructose malabsorption.[22][23]This enzyme is used in industrial settings and has been shown to produce no allergic response in humans.[22]
Products containing xylose isomerase are sold asover-the-counter(OTC)dietary supplementsto combatfructose malabsorption,under brand names includingFructaid,Fructease,Fructase,Fructose DigestandFructosin.Apart from general concerns over the effectiveness of OTC-enzymes,[24]there is currently very limited research available on Xylose-Isomerase as a dietary supplement,[25][22]with the sole scientific study[26]indicating a positive effect on malabsorption-related nausea and abdominal pain, but none on bloating.[23][25][22]This decrease in breath hydrogen excretion demonstrated in this study is a potential sign that fructose was absorbed much better.[22]However, the results of this study was not confirmed by other studies, and this study did not assess the long-term health effects and did not try to determine which patients are best suited to treatment with xylose isomerase, if at all.[22][23]
References
[edit]- ^Katz AK, Li X, Carrell HL, Hanson BL, Langan P, Coates L, et al. (May 2006)."Locating active-site hydrogen atoms in D-xylose isomerase: time-of-flight neutron diffraction".Proceedings of the National Academy of Sciences of the United States of America.103(22): 8342–8347.Bibcode:2006PNAS..103.8342K.doi:10.1073/pnas.0602598103.PMC1482496.PMID16707576.
- ^Xiao H, Gu Y, Ning Y, Yang Y, Mitchell WJ, Jiang W, et al. (November 2011)."Confirmation and elimination of xylose metabolism bottlenecks in glucose phosphoenolpyruvate-dependent phosphotransferase system-deficient Clostridium acetobutylicum for simultaneous utilization of glucose, xylose, and arabinose".Applied and Environmental Microbiology.77(22): 7886–7895.Bibcode:2011ApEnM..77.7886X.doi:10.1128/AEM.00644-11.PMC3208996.PMID21926197.
- ^Mu W, Hassanin HA, Zhou L, Jiang B (December 2018). "Chemistry Behind Rare Sugars and Bioprocessing".Journal of Agricultural and Food Chemistry.66(51): 13343–13345.doi:10.1021/acs.jafc.8b06293.PMID30543101.S2CID56145019.
- ^Mitsuhashi S, Lampen JO (October 1953)."Conversion of D-xylose to D-xylulose in extracts of Lactobacillus pentosus"(PDF).The Journal of Biological Chemistry.204(2): 1011–1018.doi:10.1016/S0021-9258(18)66103-4.PMID13117877.Archived(PDF)from the original on 2020-01-02.Retrieved2017-01-16.
- ^Schomburg D, Schomburg I (2001)."Xylose isomerase".Handbook of Enzymes.Vol. Class 5: Isomerases. New York: Springer. pp. 259–260.ISBN978-3-540-41008-9.Archivedfrom the original on 2020-07-26.Retrieved2020-07-19.
- ^Marshall RO, Kooi ER (April 1957). "Enzymatic conversion of D-glucose to D-fructose".Science.125(3249): 648–649.Bibcode:1957Sci...125..648M.doi:10.1126/science.125.3249.648.PMID13421660.
- ^abcJokela J, Pastinen O, Leisola M (2002). "Isomerization of pentose and hexose sugars by an enzyme reactor packed with cross-linked xylose isomerase crytals".Enzyme and Microbial Technology.31(1–2): 67–76.doi:10.1016/s0141-0229(02)00074-1.
- ^abcdefgWong DW (1995).Food Enzymes Structure and Mechanism.Boston, MA: Springer US.ISBN978-1-4757-2349-6.
- ^abHobbs L (2009). "21 - Sweeteners from Starch: Production, Properties and Uses". In BeMiller JN, Whistler RL (eds.).Starch: chemistry and technology(3rd ed.). London: Academic Press/Elsevier. pp.797–832.ISBN978-0-12-746275-2.
- ^"Corn syrup and its culinary uses: Is it safe to consume?".The Times of India.ISSN0971-8257.Archivedfrom the original on 2024-02-16.Retrieved2024-02-16.
- ^Beerens K, Desmet T, Soetaert W (June 2012)."Enzymes for the biocatalytic production of rare sugars".Journal of Industrial Microbiology & Biotechnology.39(6): 823–834.doi:10.1007/s10295-012-1089-x.PMID22350065.S2CID14877957.
- ^Kitahara K (1966). "Studies on Lactic Acid Bacteria".Nyusankin No Kenkyu:67~69.
- ^Buchanan RE, Gibbons NE (1974).Bergey's Manual of Determining Bacteriology(8 ed.). Baltimore: The Williams and Wilkins Co. p. 584.
- ^abcYamanaka K, Takahara N (1977)."Purification and Properties of D-Xylose Isomerase from Lactobacillus xylosus".Agric. Biol. Chem.41(10): 1909–1915.doi:10.1271/bbb1961.41.1909.
- ^"Deprecated services < EMBL-EBI".Archivedfrom the original on 2024-04-01.Retrieved2015-02-06.
- ^abcdBlow DM, Collyer CA, Goldberg JD, Smart OS (1992). "Structure and mechanism of D-xylose isomerase".Faraday Discussions.93(93): 67–73.Bibcode:1992FaDi...93...67B.doi:10.1039/fd9929300067.PMID1290940.
- ^Nam KH (2022)."Glucose Isomerase: Functions, Structures, and Applications".Applied Sciences.12:428.doi:10.3390/app12010428.
 This article incorporates text from this source, which is available under theCC BY 4.0license.
This article incorporates text from this source, which is available under theCC BY 4.0license.
- ^Qiu Y, Wu M, Bao H, Liu W, Shen Y (September 2023)."Engineering of Saccharomyces cerevisiae for co-fermentation of glucose and xylose: Current state and perspectives".Engineering Microbiology.3(3): 100084.doi:10.1016/j.engmic.2023.100084.ISSN2667-3703.
- ^Volkin DB, Klibanov AM (April 1989). "Mechanism of thermoinactivation of immobilized glucose isomerase".Biotechnology and Bioengineering.33(9): 1104–1111.doi:10.1002/bit.260330905.PMID18588027.S2CID39076432.
- ^van Maris AJ, Winkler AA, Kuyper M, de Laat WT, van Dijken JP, Pronk JT (2007)."Development of efficient xylose fermentation in Saccharomyces cerevisiae: xylose isomerase as a key component".Advances in Biochemical Engineering/Biotechnology.108:179–204.doi:10.1007/10_2007_057.ISBN978-3-540-73650-9.PMID17846724.Archivedfrom the original on 2023-12-31.Retrieved2023-12-31.
- ^Silva PC, Ceja-Navarro JA, Azevedo F, Karaoz U, Brodie EL, Johansson B (February 2021)."A novel D-xylose isomerase from the gut of the wood feeding beetle Odontotaenius disjunctus efficiently expressed in Saccharomyces cerevisiae".Scientific Reports.11(1): 4766.Bibcode:2021NatSR..11.4766S.doi:10.1038/s41598-021-83937-z.hdl:1822/72966.PMC7910561.PMID33637780.
- ^abcdefgBenardout M, Le Gresley A, ElShaer A, Wren SP (February 2022)."Fructose malabsorption: causes, diagnosis and treatment".The British Journal of Nutrition.127(4): 481–489.doi:10.1017/S0007114521001215.PMID33818329.
 This article incorporates text from this source, which is available under theCC BY 4.0license.
This article incorporates text from this source, which is available under theCC BY 4.0license.
- ^abcFernández-Bañares F (May 2022)."Carbohydrate Maldigestion and Intolerance".Nutrients.14(9): 1923.doi:10.3390/nu14091923.PMC9099680.PMID35565890.
 This article incorporates text from this source, which is available under theCC BY 4.0license.
This article incorporates text from this source, which is available under theCC BY 4.0license.
- ^Komericki P, Akkilic-Materna M, Strimitzer T, Weyermair K, Hammer HF, Aberer W (November 2012)."Oral xylose isomerase decreases breath hydrogen excretion and improves gastrointestinal symptoms in fructose malabsorption - a double-blind, placebo-controlled study".Alimentary Pharmacology & Therapeutics.36(10): 980–987.doi:10.1111/apt.12057.PMID23002720.S2CID6047336.
- ^abSingh RS, Singh T, Pandey A (2019). "Microbial Enzymes—An Overview".Advances in Enzyme Technology.pp. 1–40.doi:10.1016/B978-0-444-64114-4.00001-7.ISBN978-0-444-64114-4.
- ^Komericki P, Akkilic-Materna M, Strimitzer T, Weyermair K, Hammer HF, Aberer W (November 2012)."Oral xylose isomerase decreases breath hydrogen excretion and improves gastrointestinal symptoms in fructose malabsorption - a double-blind, placebo-controlled study".Alimentary Pharmacology & Therapeutics.36(10): 980–987.doi:10.1111/apt.12057.PMID23002720.S2CID6047336.
Further reading
[edit]- Hochster RM, Watson RW (January 1954). "Enzymatic isomerization of D-xylose to D-xylulose".Archives of Biochemistry and Biophysics.48(1): 120–129.doi:10.1016/0003-9861(54)90313-6.PMID13125579.
- Slein MW (1955). "Xylose isomerase from Pasteurella pestis, strain A-1122".J. Am. Chem. Soc.77(6): 1663–1667.doi:10.1021/ja01611a074.
- Yamanaka K (March 1968). "Purification, crystallization and properties of the D-xylose isomerase from Lactobacillus brevis".Biochimica et Biophysica Acta (BBA) - Enzymology.151(3): 670–680.doi:10.1016/0005-2744(68)90015-6.PMID5646045.
