Irsogladine
Appearance
| |
| Names | |
|---|---|
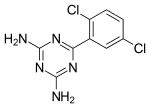
| Preferred IUPAC name
6-(2,5-Dichlorophenyl)1,3,5-triazine-2,4-diamine | |
| Other names
DCPDAT, Dicloguamine, MN-1695.
| |
| Identifiers | |
| ChemSpider | |
PubChemCID
|
|
| UNII | |
CompTox Dashboard(EPA)
|
|
| Properties | |
| C9H7Cl2N5 | |
| Molar mass | 256.09g·mol−1 |
| Pharmacology | |
| A02BX16(WHO) | |
Except where otherwise noted, data are given for materials in theirstandard state(at 25 °C [77 °F], 100 kPa).
| |
Irsogladineis aphosphodiesterase inhibitor.[1]
Antiulcerdrug.
Synthesis
[edit]
2,4-Dichloro-6-(2,5-dichlorophenyl)-1,3,5-triazine [61479-79-6] (1) (2) Dicyandiamide[461-58-5] (3) 2,5-Dichlorobenzonitrile[21663-61-6] (4)
References
[edit]- ^Satoh, H; Amagase, K; Takeuchi, K (2013). "Mucosal protective agents prevent exacerbation of NSAID-induced small intestinal lesions caused by antisecretory drugs in rats".The Journal of Pharmacology and Experimental Therapeutics.348(2): 227–35.doi:10.1124/jpet.113.208991.PMID24254524.S2CID23740642.
- ^Serradell, MN; Blancafort, P.; Castaer, J.; MN-1695. Drugs Fut 1983, 8, 6, 502.
- ^Ueda F, Aratani S, Mimura K, Kimura K, Nomura A, Enomoto H. Effect of 2,4-diamino-6-(2,5-dichlorophenyl)-s-triazine maleate (MN-1695) on gastric ulcers and gastric secretion in experimental animals. Arzneimittelforschung. 1984;34(4):474-7. PMID: 654016.
- ^DE2506814 idem Hiromu Murai, 6 More »,U.S. patent 3,966,728(1976 to Nippon Shinyaku Co., Ltd.).
