Luzindole
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChemCID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| CompTox Dashboard(EPA) | |
| Chemical and physical data | |
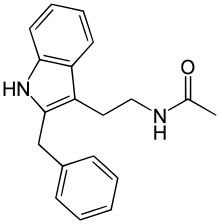
| Formula | C19H20N2O |
| Molar mass | 292.382g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Luzindole(N-0774), (N-acetyl-2-benzyltryptamine), is adrugused inscientific researchto study the role ofmelatoninin the body. Luzindole acts as aselectivemelatonin receptorantagonist,[1]with approximately 11- to 25-fold greateraffinityfor theMT2over theMT1receptor.[2][3]In animal studies, it has been observed to disrupt thecircadian rhythmas well as produceantidepressanteffects.[2][4]
Synthesis[edit]
Although the "hydrogen bomb" method was reported as 54% yield by Dubococvich, Boehringer Sohn achieved 96% for this step. The difference is that B.I. conducted their hydrogenation under normal pressure at 50°C for 5 hours, whereas Dubocovich conducted theirs at 100 lbs/in2 hydrogen heated to 35°C. This proves that the hydrogenation step proceeds favorably under milder conditions.

ThePictet–Spengler reactionbetween tryptamine [61-54-1] (1) and benzaldehyde gives 1-Phenyl-tetrahydrocarboline [3790-45-2] (2). Catalytic hydrogenation leads to 2-Benzyltryptamine [22294-23-1] (3). Acylation with acetic anhydride only gave 21% yield of Luzindole (4).

2-iodoaniline [615-43-0] (1) Propargylbenzene [10147-11-2] (2) 2-(3-phenylprop-1-ynyl)aniline, PC85868179 (3) 2-benzylindole [3377-72-8] (4) 1-Dimethylamino-2-nitroethylene [1190-92-7] (5) (6)
One potLuzindole synthesis:[9]
References[edit]
- ^Dubocovich ML (September 1988)."Luzindole (N-0774): a novel melatonin receptor antagonist".The Journal of Pharmacology and Experimental Therapeutics.246(3): 902–10.PMID2843633.
- ^abDubocovich ML, Yun K, Al-Ghoul WM, Benloucif S, Masana MI (September 1998)."Selective MT2 melatonin receptor antagonists block melatonin-mediated phase advances of circadian rhythms".The FASEB Journal.12(12): 1211–20.doi:10.1096/fasebj.12.12.1211.PMID9737724.S2CID566199.
- ^Browning C, Beresford I, Fraser N, Giles H (March 2000)."Pharmacological characterization of human recombinant melatonin mt(1) and MT(2) receptors".British Journal of Pharmacology.129(5): 877–86.doi:10.1038/sj.bjp.0703130.PMC1571913.PMID10696085.
- ^Dubocovich ML, Mogilnicka E, Areso PM (July 1990). "Antidepressant-like activity of the melatonin receptor antagonist, luzindole (N-0774), in the mouse behavioral despair test".European Journal of Pharmacology.182(2): 313–25.doi:10.1016/0014-2999(90)90290-M.PMID2168835.
- ^Margarita L. Dubocovich, et al. WO1989001472A1 ().
- ^Margarita L. Dubocovich, et al.,U.S. patent 5,283,343(1994 to Discovery Therapeutics Inc).
- ^Schroeder Dr Hans-D, et al. DE1445516 (1968 to CH Boehringer Sohn AG and Co KG).
- ^Tsotinis, Andrew; Afroudakis, Pandelis (2008). "Melatonin Receptor Antagonist Luzindole: A Facile New Synthesis". Letters in Organic Chemistry. 5 (6): 507–509. doi:10.2174/157017808785740561. ISSN 1570-1786.
- ^Righi, Marika; Topi, Francesca; Bartolucci, Silvia; Bedini, Annalida; Piersanti, Giovanni; Spadoni, Gilberto (2012). "Synthesis of Tryptamine Derivatives via a Direct, One-Pot Reductive Alkylation of Indoles". The Journal of Organic Chemistry. 77 (14): 6351–6357. doi:10.1021/jo3010028.
