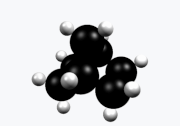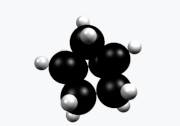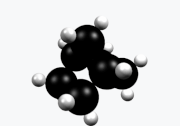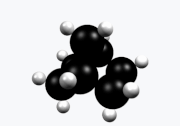
Norbornane
Appearance

| |

| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
Bicyclo[2.2.1]heptane | |
| Other names
norcamphane, norbornylane
| |
| Identifiers | |
3D model (JSmol)
|
|
| 1900379 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.005.452 |
| EC Number |
|
PubChemCID
|
|
| UNII | |
CompTox Dashboard(EPA)
|
|
| |
| |
| Properties | |
| C7H12 | |
| Molar mass | 96.17 g mol−1 |
| Appearance | white volatile solid |
| Melting point | 85 to 88 °C (185 to 190 °F; 358 to 361 K) |
Except where otherwise noted, data are given for materials in theirstandard state(at 25 °C [77 °F], 100 kPa).
| |
Norbornane(also known asbicyclo[2.2.1]heptane) is anorganic compoundand asaturatedhydrocarbonwithchemical formulaC7H12.It is acrystallinecompound with amelting pointof 88°C.The carbon skeleton is derived fromcyclohexanering with amethylene bridgein the 1,4- position, and is a bridgedbicycliccompound. The compound is a prototype of a class of strained bicyclic hydrocarbons.
The compound was originally synthesized by reduction ofnorcamphor.[1]
The name norbornane is derived frombornane,which is 1,7,7-trimethylnorbornane, being a derivative ofcamphor(bornanone). The prefixnorrefers to the stripping of the methyl groups from the parent molecule bornane.
See also
[edit]- 2-Norbornyl cation
- Norbornene
- Norbornadiene
- Bornane
- endo-Norborneol
- exo-Norborneol
- Norcamphor,the ketone derivative of norbornane
References
[edit]- ^Komppa, Gust.; Beckmann, Siegfried (1934). "Der Grundkörper der Camphergruppe, das Bicyclo-[1.2.2]-heptan, und die stereoisomeren Norborneole".Naturwissenschaften.22:171.doi:10.1007/BF01496254.
