Holmium
Erscheinungsbild
| Eegenskapen | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Algemian | ||||||||||||||||||||||||||||||||||||||||
| Nööm, Symbool, Numer | Holmium, Ho, 67 | |||||||||||||||||||||||||||||||||||||||
| Seerie | Lantanoid | |||||||||||||||||||||||||||||||||||||||
| Skööl, Periode, Blook | La,6,f | |||||||||||||||||||||||||||||||||||||||
| Klöör, Skak | salwern witj | |||||||||||||||||||||||||||||||||||||||
| CAS-Numer | 7440-60-0 | |||||||||||||||||||||||||||||||||||||||
| Uundial | 1,1 ppm[1] | |||||||||||||||||||||||||||||||||||||||
| Atomaar[2] | ||||||||||||||||||||||||||||||||||||||||
| Atoommase | 164,93033(2)[3][4]u | |||||||||||||||||||||||||||||||||||||||
| Atoomraadius (bereegent) | 175 (226)pm | |||||||||||||||||||||||||||||||||||||||
| Kovalent-Raadius | 192 pm | |||||||||||||||||||||||||||||||||||||||
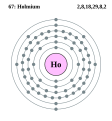
| Elektroonen | [Xe] 4f116s2 | |||||||||||||||||||||||||||||||||||||||
| 1. Ionisiarang | 581,0kJ/mol | |||||||||||||||||||||||||||||||||||||||
| 2. Ionisiarang | 1140 kJ/mol | |||||||||||||||||||||||||||||||||||||||
| 3. Ionisiarang | 2204 kJ/mol | |||||||||||||||||||||||||||||||||||||||
| Füsikaalisk[2] | ||||||||||||||||||||||||||||||||||||||||
| Tustant | fääst | |||||||||||||||||||||||||||||||||||||||
| Kristal | hexagonaal | |||||||||||||||||||||||||||||||||||||||
| Sachthaid | 8,78 g/cm3(25°C)[5] | |||||||||||||||||||||||||||||||||||||||
| Magnetismus | paramagneetisk(Χm= 0,049)[6] | |||||||||||||||||||||||||||||||||||||||
| Smoltponkt | 1734K(1461 °C) | |||||||||||||||||||||||||||||||||||||||
| Köögponkt | 2873 K[7]K(2600 °C) | |||||||||||||||||||||||||||||||||||||||
| Molaar Rüm | 18,74 · 10−6m3/mol | |||||||||||||||||||||||||||||||||||||||
| Dampwaremk | 251 kJ/mol[7]kJ/mol | |||||||||||||||||||||||||||||||||||||||
| Smoltwaremk | 17 kJ/mol | |||||||||||||||||||||||||||||||||||||||
| Faard faan a tuun | 2760 m/s | |||||||||||||||||||||||||||||||||||||||
| Elektrisk struumfeerang | 1,23 · 106A/(V · m) | |||||||||||||||||||||||||||||||||||||||
| Waremkfeerang | 16W/(m · K) | |||||||||||||||||||||||||||||||||||||||
| Cheemisk[2] | ||||||||||||||||||||||||||||||||||||||||
| Oksidatsionstustant | 3 | |||||||||||||||||||||||||||||||||||||||
| Sürofbaasisk | swaakbaasisk | |||||||||||||||||||||||||||||||||||||||
| Normoolpotentiaal | −2,33V(Ho3++ 3 e−→ Ho) | |||||||||||||||||||||||||||||||||||||||
| Elektronegatiwiteet | 1,23 (Pauling-Skala) | |||||||||||||||||||||||||||||||||||||||
| Isotoopen | ||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||
| Muar isotoopen biList faan isotoopen | ||||||||||||||||||||||||||||||||||||||||
| Seekerhaid | ||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||
| Miast wurdSI-ianhaiden brükt. | ||||||||||||||||||||||||||||||||||||||||
Holmiumas encheemisk elementmä det ufkörtangHoan detatoomnumer67.Hat hiart tu alantanoiden.
Bilen
[Bewerke|Kweltekst bewerke]-
Elektroonenskel
-
Rian holmium
-
Holmium
Luke uk diar
[Bewerke|Kweltekst bewerke]Kwelen
[Bewerke|Kweltekst bewerke]- ↑Harry H. Binder:Lexikon der chemischen Elemente.S. Hirzel Verlag, Stuttgart 1999,ISBN 3-7776-0736-3.
- ↑A taalen för't infobox kem miast faanwww.webelements.com (Holmium).
- ↑IUPAC, Standard Atomic Weights Revised v2(Memento faan di 3. Marts 2016 uun'tInternet Archive).
- ↑CIAAW, Standard Atomic Weights Revised 2013.
- ↑N. N. Greenwood, A. Earnshaw:Chemie der Elemente.1. aplaag. VCH, Weinheim 1988,ISBN 3-527-26169-9,S. 1579.
- ↑Robert C. Weast (Hrsg.):CRC Handbook of Chemistry and Physics.CRC (Chemical Rubber Publishing Company), Boca Raton 1990,ISBN 0-8493-0470-9,S. E-129 bit E-145. Wäärser diar uun g/mol an uuncgs-ianhaiden. Heer amreegent tuSI-wäärs.
- ↑7,07,1Yiming Zhang, Julian R. G. Evans, Shoufeng Yang:Corrected Values for Boiling Points and Enthalpies of Vaporization of Elements in Handbooks.Uun:Journal of Chemical & Engineering Data.56, 2011, S. 328–337,doi:10.1021/je1011086.
- ↑8,08,1Iindraanj tuHolmium, Pulveruun't GESTIS-dootenbeenk faan'tIFA,ufrepen di 26. April 2017(mä JavaScript).